GUIDANCE DOCUMENT
Q8, Q9, & Q10 Questions and Answers -- Appendix: Q&As from Training Sessions (Q8, Q9, & Q10 Points to Consider) August 2012
Guidance for Industry
I. INTRODUCTION [2]
The ICH Quality Implementation Working Group (Q-IWG) has prepared this Points to Consider document covering topics relevant to the implementation of ICH Q8(R2), Q9, and Q10, which supplement the existing guidance Q8, Q9, and Q10 Questions & Answers [3] and workshop training materials [4] already produced by this group. They should be considered all together.
The points to consider are based on questions raised during the ICH Q-IWG training workshop sessions in the three regions. The points to consider are not intended to be new guidance. They are intended to provide clarity to both industry and regulators and to facilitate the preparation, assessment, and inspection related to applications filed for marketing authorizations.
The development approach should be adapted based on the complexity and specificity of product and process; therefore, applicants are encouraged to contact regulatory authorities regarding questions related to specific information to be included in their application.
Using the Quality by Design (QbD) approach does not change regional regulatory requirements but can provide opportunities for more flexible approaches to meet them. In all cases, good manufacturing practice (GMP) compliance is expected.
II. WHAT ARE THE POINTS TO CONSIDER FOR CRITICALITY OF QUALITY ATTRIBUTES AND PROCESS PARAMETERS
Scientific rationale and quality risk management (QRM) processes are used to reach a conclusion on what are critical quality attributes (CQAs) and critical process parameters (CPPs) for a given product and process
The quality target product profile (QTPP) describes the design criteria for the product, and should therefore form the basis for development of the CQAs, CPPs, and control strategy.
The information developed to determine CQAs and CPPs will help to:
• Develop control strategy
• Ensure quality of the product throughout the product lifecycle
• Increase product and process knowledge
• Increase transparency and understanding for regulators and industry
• Evaluate changes
A. Considerations for Establishing CQAs and CPPs
The introduction of ICH Q9 states that “…the protection of the patient by managing the risk to quality should be considered of prime importance.” The QTPP provides an understanding of what will ensure the quality, safety, and efficacy of a specific product for the patient and is a starting point for identifying the CQAs.
As part of risk assessment, risk analysis, as defined by ICH Q9 is “the qualitative or quantitative process of linking the likelihood of occurrence and severity of harm. In some risk management tools, the ability to detect the harm (detectability) also factors in the estimation of risk.”
Relationship between risk and criticality:
• Risk includes severity of harm, probability of occurrence, and detectability, and therefore the level of risk can change as a result of risk management.
• Quality attribute criticality is primarily based upon severity of harm and does not change as a result of risk management.
• Process parameter criticality is linked to the parameter’s effect on any critical quality attribute. It is based on the probability of occurrence and detectability and therefore can change as a result of risk management.
Considerations for identifying and documenting CQAs can include the:
• Severity of harm (safety and efficacy) before taking into account risk control and the rationale for distinguishing CQAs from other quality attributes;
• Link to the patient as described in the QTPP;
• Basis on which the CQAs have been developed (e.g., prior knowledge, scientific first principles, and experimentation); and
• Inter-dependencies of the different CQAs.
Considerations for identifying and documenting CPPs can include the:
• Risk assessment and experimentation to establish the linkage between potential CPPs and CQAs;
• Basis on which the CPPs have been identified (e.g., prior knowledge, scientific first principles, QRM, design of experiment (DoE), and other appropriate experimentation);
• Interdependencies of the different CPPs; and
• Selected control strategy and the residual risk.
CQAs and CPPs can evolve throughout the product lifecycle, for example:
• Change of manufacturing process (e.g., change of synthetic route) and
• Subsequent knowledge gained throughout the lifecycle (e.g., raw material variability, pharmacovigilance, clinical trial experience, and product complaints).
B. Relationship of Criticality to Control Strategy
The identification and linkage of the CQAs and CPPs should be considered when designing the control strategy. A well-developed control strategy will reduce risk but does not change the criticality of attributes.
The control strategy plays a key role in ensuring that the CQAs are met and, hence, that the QTPP is realized.
III. WHAT ARE THE POINTS TO CONSIDER FOR CONTROL STRATEGY
A. Lifecycle of the Control Strategy
The lifecycle of the control strategy is supported by pharmaceutical development, quality risk management (QRM), and the pharmaceutical quality system (PQS), as described in ICH Q8(R2), Q9, and Q10.
The following points can be considered:
• Development of control strategy:
• The control strategy is generally developed and initially implemented for production of clinical trial materials. It can be refined for use in commercial manufacture as new knowledge is gained. Changes could include acceptance criteria, analytical methodology, or the points of control (e.g., introduction of real-time release testing).
• Additional emphasis on process controls should be considered in cases where products cannot be well-characterized and/or quality attributes might not be readily measurable due to limitations of testing or detectability (e.g., microbial load/sterility).
• Continual improvement of the control strategy:
• Consideration should be given to improving the control strategy over the lifecycle (e.g., in response to assessment of data trends over time and other knowledge gained).
• Continuous process verification is one approach that enables a company to monitor the process and make adjustments to the process and/or the control strategy, as appropriate.
• When multivariate prediction models are used, systems that maintain and update the models help to assure the continued suitability of the model within the control strategy.
• Change management of the control strategy:
• Attention should be given to outsourced activities to ensure all changes are communicated and managed.
• The regulatory action appropriate for different types of changes should be handled in accordance with the regional regulatory requirements.
• Different control strategies for the same product:
• Different control strategies could be applied at different sites or when using different technologies for the same product at the same site.
• Differences might be due to equipment, facilities, systems, business requirements (e.g., confidentiality issues, vendor capabilities at outsourced manufacturers) or as a result of regulatory assessment/inspection outcomes.
• The applicant should consider the impact of the control strategy implemented on the residual risk and the batch release process.
• Knowledge management:
• Knowledge management is an important factor in assuring the ongoing effectiveness of the control strategy.
• For contract manufacturing, knowledge transfer in both directions between the parties should be considered, particularly for model maintenance and/or updates, application of design space, and control strategies incorporating real-time release testing.
B. Suitability of Control Strategy at Different Scales
• Management of risk on scale-up:
• Risk associated with scale-up should be considered in control strategy development to maximize the probability of effectiveness at scale. The design and need for scale-up studies can depend on the development approach used and knowledge available.
• A risk-based approach can be applied to the assessment of suitability of a control strategy across different scales. QRM tools can be used to guide these activities. This assessment might include risks from processing equipment, facility environmental controls, personnel capability, experiences with technologies, and historical experience (prior knowledge). See the ICH Q-IWG case study for examples.
• Scale-up considerations for elements of Control Strategy:
• Complexity of product and process
• Differences in manufacturing equipment, facilities and/or sites
• Raw materials:
- Differences in raw material quality due to source or batch-to-batch
variability
- Impact of such differences on process controls and quality attributes
• Process parameters:
- Confirmation or optimization
- Confirmation of the design space(s), if used
• In-process controls:
- Point of control
- Optimization of control methods
- Optimization and/or updating of models, if used
• Product specification:
- Verification of the link to QTPP
- Confirmation of specifications (i.e., methods and acceptance criteria)
- Confirmation of real-time release testing (RTRT), if used
C. Specifications and Certificate of Analysis (CoA) for Real-Time Release Testing (RTRT)
The purpose of specifications and CoAs remains the same in the case of RTRT, but the way to develop them is different. Real-time release tests(RTRT) are considered to be specification testing methods and follow the established regional regulatory requirements for release specifications (as interpreted in e.g., ICH Q6A and ICH Q6B) together with other regional regulatory requirements (e.g., formats, GMP, batch acceptance decisions).
The use of RTRT has been addressed (see ICH Q8(R2), section II.E (2.5); ICH Q8, Q9, and Q10 Q&As, section II.B (2.2)). The following points should be considered when developing a specification and CoA for RTRT:
• Quality attributes:
• Not all CQAs need to be included in the specification.
• The attribute to be measured (e.g., surrogate for a CQA) can depend on the point of testing and/or control (e.g., materials, process steps, process parameters).
• Linking of the measured attribute to CQA and QTPP
• Methods of control:
• The type of control used (e.g., models, process analytical technology (PAT), test of isolated material, end product test, stability and regulatory test)
• Reference to the testing method used, if relevant
• Validation of control method
• Acceptance criteria:
• Acceptance criteria at control point
• Criteria for stability and regulatory testing
• CoA elements:
• Reported results (e.g., values calculated from models, established calibrations, and actual test results)
• Acceptance criteria related to the method used
• Method references
D. Process for a Batch Release Decision
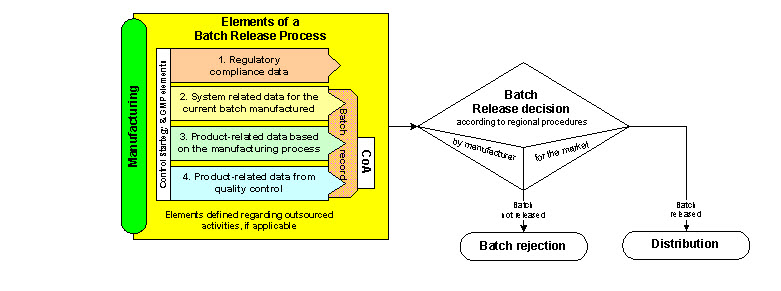
Different development approaches lead to different control strategies. Regardless of the control strategy, the batch release process should be followed. For a batch release decision, several elements should be considered. See in the figure below an illustration of the elements of the batch release process leading to the batch release decision.
1. Regulatory compliance data:
There are regional differences in the regulation of batch release across the ICH regions (e.g., Qualified Person (EU), Good Quality Practice (Japan), Head of Quality Unit (U.S.)) and the manufacturing licensing procedure. The PQS facilitates implementing and managing control strategy and batch release, notably through elements of a global approach (corporate/site/contractor). The PQS elements also facilitate regulatory compliance (e.g., changes that call for variation of the marketing authorization), including changes at manufacturing sites (e.g., changes regarding facilities, utilities, and equipment).
2. System-related data for the current batch manufactured (e.g., environmental, facility, utilities and equipment)
In the enhanced approach, there is an increased focus on process monitoring, which can provide the opportunity to perform continuous process verification. Any deviation or atypical event that occurs during manufacturing (e.g., involving the manufacturing process, facility, personnel, testing) should be recorded and assessed, properly handled under the PQS (including corrective and preventive action (CAPA)), and closed out prior to release.
3. Product-related data based on the manufacturing process
Elements of the control strategy are defined and proposed in the marketing authorization dossier and agreed to by the regulators. Manufacturers should define, manage, and monitor product-related data from batches manufactured according to the control strategy. These will be regularly assessed and reviewed during audits and inspections.
4. Product-related data from quality control
Results from end product testing and/or RTRT provide data based on which a CoA can be issued, in compliance with the specification as part of the release decision.
The batch release process leading to the batch release decision can be performed by more than one quality individual depending on the regional regulatory requirements and company policy:
• Batch release by manufacturer or contractor for internal purposes
• Batch release by manufacturing authorization holder for the market
IV. WHAT ARE THE POINTS TO CONSIDER FOR LEVEL OF DOCUMENTATION IN ENHANCED (QBD) REGULATORY SUBMISSIONS
This document is intended to provide suggestions on the type of information and the level of documentation that is appropriate to support a proposal for enhanced (QbD) approach. The type of information, as suggested in this document, is considered supportive and is intended to facilitate assessment and inspection without increasing the regulatory requirement. Submitted information should be organized in a clear manner and provide the regulators with sufficient understanding of the company’s development approach; this information will be important to the evaluation of the proposed control strategy. Companies might consider, especially for QbD-containing submissions, an internal peer review process to assure quality, clarity, and adequacy of the regulatory submission.
For submissions containing QbD elements (e.g., RTRT, design space), it is helpful for regulators to have a statement by the applicant describing the proposed regulatory outcome and expectations.
It is important to realize that not all the studies performed and/or data generated during product development need to be submitted. However, sufficient supporting information and data should be submitted in the application to address the following:
• The scientific justification of the proposed control strategy;
• The scientific rationale for the studies conducted;
• A concise description of methodologies used to conduct these studies and to analyze the generated data; and
• The summary of results and conclusions drawn from these studies.
The following sections include examples of background information that can be considered by both companies and regulatory authorities to assure scientific risk-based regulatory decisions.
A. Risk Management Methodologies
Following determination of the quality target product profile (QTPP) of the product under development, the applicant can use quality risk management (QRM, ICH Q9) tools to rank and select quality attributes (including material attributes) and/or process parameters that should be further evaluated and/or controlled within appropriate ranges to ensure the desired product quality. The applicant should consider providing information of sufficient detail to demonstrate how the conclusions were reached, which can include:
• The scientific rationale for designation of QTPP and identification of corresponding CQAs (critical quality attributes);
• Material attributes, process parameters, and prior knowledge that were considered during risk assessment, preferably provided in a concise/tabulated form;
• Relevant known risk factors (e.g., degradation, solubility);
• The scientific rationale and basis for the risk assessment as part of risk management and experiments that determined the final criticality of quality attributes and process parameters;
• Identification of potential residual risk that might remain after the implementation of the proposed control strategy (e.g., movements to commercially unverified areas of design space) and discussion of approaches for managing the residual risk;
• A list of critical and other quality attributes and process parameters;
• The linkage between CPPs , CQAs, and the QTPP; and
• Comment on the impact of the following on risk assessment: (a) interaction of attributes and process parameters, (b) effect of equipment and scale.
The factors to be studied in a DoE could come from the risk assessment exercise or prior knowledge. Inclusion of a full statistical evaluation of the DoEs performed at early development stages (e.g., screening) is not expected. A summary table of the factors and ranges studied and the conclusions reached will be helpful. For DoEs involving single- or multiple-unit operations that are used to establish CPPs and/or to define a Design Space (DS), the inclusion of the following information in the submission will greatly facilitate assessment by the regulators:
• Rationale for selection of DoE variables (including ranges) that would be chosen by risk assessment (e.g., consideration of the potential interactions with other variables).
• Any evidence of variability in raw materials (e.g., drug substance and/or excipients) that would have an impact on predictions made from DoE studies.
• Listing of the parameters that would be kept constant during the DoEs and their respective values, including comments on the impact of scale on these parameters.
• Type of experimental design used and a justification of its appropriateness, including the power of the design.
• Factors under study and their ranges can be presented in a tabular format. Submitters should indicate if the factors are expected to be scale-dependent.
• Reference to the type of analytical methods (e.g., high-pressure liquid chromatography (HPLC), near infrared (NIR)) used for the evaluation of the data and their suitability for their intended use (e.g., specificity, detection limit).
• Results and statistical analysis of DoE data showing the statistical significance of the factors and their interactions, including predictions made from DoE studies relevant to scale and equipment differences.
C. Manufacturing Process Description
While preparing regulatory submissions, applicants should consider:
• Regional regulatory requirements with regard to the level of detail in describing manufacturing processes.
• Describing the proposed design space, including critical and other parameters studied, and its role in the development of the control strategy.
• Manufacturing changes should be managed in accordance with regional regulatory requirements. Where relevant, applicants can also consider submitting postapproval change management plans or protocols to manage postapproval manufacturing changes based on regional requirements.
V. WHAT ARE THE POINTS TO CONSIDER FOR ROLE OF MODELS IN QUALITY BY DESIGN (QBD)
A model is a simplified representation of a system using mathematical terms. Models can enhance scientific understanding and possibly predict the behavior of a system under a set of conditions. Mathematical models can be used at every stage of development and manufacturing. They can be derived from first principles reflecting physical laws (such as mass balance, energy balance, and heat transfer relations), or from data, or from a combination of the two. There are many types of models and the selected one will depend on the existing knowledge about the system, the data available, and the objective of the study. This document is intended to highlight some points to consider when developing and implementing mathematical models during pharmaceutical product development, manufacturing and throughout the product lifecycle. Other approaches not described in this document can also be used.
Models can be categorized in multiple ways. The categorization approaches used throughout this document are intended to facilitate the use of models across the lifecycle, including development, manufacturing, control, and regulatory processes.
For the purposes of regulatory submissions, an important factor to consider is the model’s contribution in assuring the quality of the product. The level of oversight should be commensurate with the level of risk associated with the use of the specific model. The following is an example of such a categorization:
1. Low-Impact Models:
These models are typically used to support product and/or process development (e.g., formulation optimization).
2. Medium-Impact Models:
Such models can be useful in assuring quality of the product but are not the sole indicators of product quality (e.g., most design space models, many in-process controls).
3. High-Impact Models:
A model can be considered high impact if prediction from the model is a significant indicator of quality of the product (e.g., a chemometric model for product assay, a surrogate model for dissolution).
For the purpose of implementation, models can also be categorized on the basis of the intended outcome of the model. Within each of these categories, models can be further classified as low, medium or high, on the basis of their impact in assuring product quality.
Some examples of different categories based on intended use are:
• Models for supporting process design:
This category of models includes (but is not limited to) models for formulation optimization, process optimization (e.g., reaction kinetics model), design space determination, and scale-up. Models within this category can have different levels of impact. For example, a model for design space determination would generally be considered a medium-impact model, while a model for formulation optimization would be considered a low-impact model.
• Models for supporting analytical procedures:
In general, this category includes empirical (i.e., chemometric) models based on data generated by various Process Analytical Technology (PAT)-based methods, for example, a calibration model associated with a near infrared (NIR)-based method. Models for supporting analytical procedures can have various impacts depending on the use of the analytical method. For example, if the method is used for release testing, then the model should be high-impact.
• Models for process monitoring and control:
This category includes, but is not limited to:
• Univariate Statistical Process Control (SPC) or Multivariate Statistical Process Control (MSPC)-based models:
These models are used to detect special cause variability; the model is usually derived and the limits are determined using batches manufactured within the target conditions. If an MSPC model is used for continuous process verification along with a traditional method for release testing, then the MSPC model would likely be classified as a medium-impact model. However, if an MSPC model is used to support a surrogate for a traditional release testing method in an RTRT approach, then the model would likely be classified as a high-impact model.
• Models used for process control (e.g., feed forward or feedback).
Data-driven models should be developed through appropriately designed experiments. These models are typically medium-impact or high-impact. For example, a feed forward model to adjust compression parameters on the basis of incoming material attributes could be classified as a medium-impact model.
B. Developing and Implementing Models
The following steps, if applicable, can be followed in a sequential manner, but occasionally, it might be appropriate to repeat an earlier step, thus imparting an iterative nature to this process. The overall steps are:
1. Defining the purpose of the model.
2. Deciding on the type of modeling approach (e.g. mechanistic or empirical) and the possible experimental/sampling methodology to be used to support the model development.
3. Selecting variables for the model; this is typically based on risk assessment, underlying physicochemical phenomena, inherent process knowledge, and prior experience.
4. Understanding the limitations of the model assumptions to:
a. Correctly design any appropriate experiments;
b. Interpret the model results; and
c. Include appropriate risk-reduction strategies.
5. Collecting experimental data to support model development. These data can be collected at laboratory, pilot, or commercial scale, depending on the nature of the model. It is important to ensure that variable ranges evaluated during model development are representative of conditions that would be expected during operation.
6. Developing model equations and estimating parameters, based on a scientific understanding of the process and collected experimental data.
7. Validating the model, as appropriate (see section V.C (5.3)).
8. In certain cases, evaluating the impact of uncertainty in model prediction on product quality and, if appropriate, defining an approach to reduce associated residual risk (e.g., by incorporating appropriate control strategies (this can apply to high-impact and medium-impact models)).
9. Documenting the outcome of model development, including model assumptions, and developing plans for verification and update of the model throughout the lifecycle of the product. The level of documentation would be dependent on the impact of the model (see section V.D (5.4)).
C. Model Validation and Model Verification During the Lifecycle
Model validation is an essential part of model development and implementation. Once a model is developed and implemented, verification continues throughout the lifecycle of the product.
The following elements can be considered for model validation and verification and generally are appropriate for high-impact models. In the case of well-established first principles-driven models, prior knowledge can be leveraged to support model validation and verification, if applicable. The applicability of the elements listed below for medium-impact or low-impact models can be considered on a case-by-case basis.
• Setting acceptance criteria for the model relevant to the purpose of the model and to its expected performance. In setting the acceptance criteria, variability in sampling procedure (e.g., for blending) could also be considered. In situations where the model is to be used to support a surrogate for a traditional release testing method, the accuracy of the model performance versus the reference method could be considered. For example, a multivariate model (e.g. a partial least squares (PLS) model), when appropriate, can be used as a surrogate for traditional dissolution testing. In this case, the PLS model should be developed in terms of in-process parameters and material attributes and can be used to predict dissolution. One of the ways to validate and verify model performance in this case would be to compare accuracy of prediction of the PLS model with the reference method (e.g., a traditional dissolution method).
• Comparison of the accuracy of calibration versus the accuracy of prediction. This can often be approached through internal cross-validation techniques using the same data as the calibration data set.
• Validating the model using an external data set (i.e., a data set from experiments/batches not used for model-building).
• It can be beneficial to verify the prediction accuracy of the model by parallel testing with the reference method during the initial stage of model implementation, and this testing can be repeated throughout the lifecycle, as appropriate. If models are used to support a design space at commercial scale or are part of the control strategy, it is important to verify the model at commercial scale—for example, if a calibration model associated with an NIR-based method is developed at the laboratory scale and the method is then transferred to and used in commercial scale. In addition, the data sets used for calibration, internal validation, and external validation should take into account the variability anticipated in future routine production (e.g., a change in the source of raw material that might impact NIR prediction). Low-impact models typically do not call for verification.
Approaches for model verification can be documented according to the PQS of the company and can include the following: a risk-based frequency of comparing the model’s prediction with that of the reference method, triggers for model updates (e.g., because of changes in raw materials or equipment), procedures for handling model-predicted Out of Specification (OOS) results, periodic evaluations, and approaches to model recalibration.
D. Documentation of Model-related Information
The level of detail for describing a model in a regulatory submission is dependent on the impact of its implementation in assuring the quality of the product. For the various types of models, the applicant can consider including:
1. Low-Impact Models:
A discussion of how the models were used to make decisions during process development.
2. Medium-Impact Models:
Model assumptions, a tabular or graphical summary of model inputs and outputs, relevant model equations (e.g., for mechanistic models) either in the submission or via a reference, statistical analysis where appropriate, a comparison of model prediction with measured data, and a discussion of how the other elements in the control strategy help to mitigate uncertainty in the model, if appropriate.
3. High-Impact Models:
Data and/or prior knowledge (e.g., for established first principles-driven models) such as model assumptions, appropriateness of the sample size, number and distribution of samples, data pretreatment, justification for variable selection, model inputs and outputs, model equations, statistical analysis of data showing fit and prediction ability, rationale for setting of model acceptance criteria, model validation (internal and external), and a general discussion of approaches for model verification during the lifecycle.
VI. WHAT ARE THE POINTS TO CONSIDER FOR DESIGN SPACE
A. Development of Design Space
A design space can be updated over the lifecycle as additional knowledge is gained. Risk assessments, as part of the risk management process, help steer the focus of development studies and define the design space. Operating within the design space is part of the control strategy. The design space associated with the control strategy ensures that the manufacturing process produces a product that meets the Quality Target Product Profile (QTPP) and Critical Quality Attributes (CQAs).
Since design spaces are typically developed at small scale, an effective control strategy helps manage potential residual risk after development and implementation. When developing a design space for a single-unit operation, the context of the overall manufacturing process can be considered, particularly immediate upstream and downstream steps that could interact with that unit operation. Potential linkages to CQAs should be evaluated in design space development.
In developing design spaces for existing products, multivariate models can be used for retrospective evaluation of historical production data. The level of variability present in the historical data will influence the ability to develop a design space, and additional studies might be appropriate.
Design spaces can be based on scientific first principles and/or empirical models. An appropriate statistical design of experiments incorporates a level of confidence that applies to the entire design space, including the edges of an approved design space. However, when operating the process near the edges of the design space, the risk of excursions from the design space could be higher because of normal process variation (common cause variation). The control strategy helps manage residual risk associated with the chosen point of operation within the design space. When changes are made (e.g., process, equipment, raw material suppliers), results of risk review can provide information regarding additional studies and/or testing that might verify the continued applicability of the design space and associated manufacturing steps after the change.
Capturing development knowledge and understanding contributes to design space implementation and continual improvement. Different approaches can be considered when implementing a design space (e.g., process ranges, mathematical expressions, or feedback controls to adjust parameters during processing (see also Figure 1d in ICH Q8(R2)). The chosen approach would be reflected in the control strategy to assure the inputs and process stay within the design space.
B. Verification and Scale-up of Design Space
Although the entire design space does not have to be reestablished (e.g., DoE) at commercial scale, design spaces should be initially verified as suitable prior to commercial manufacturing. Design space verification should not be confused with process validation. However, it might be possible to conduct verification studies of the performance of the design space scale-dependent parameters as part of process validation. Design space verification includes monitoring or testing of CQAs that are influenced by scale-dependent parameters. Additional verification of a design space might be triggered by changes (e.g., site, scale, or equipment). Additional verification is typically guided by the results of risk assessments of the potential impacts of the change(s) on design space.
A risk-based approach can be applied to determine the design of any appropriate studies for assessment of the suitability of a design space across different scales. Prior knowledge and first principles, including simulation models and equipment scale-up factors, can be used to predict scale-independent parameters. Experimental studies could help verify these predictions.
C. Documentation of Design Space
Information on design space can be accommodated in the common technical document (CTD) in different presentation formats. Some examples of format and location in the document are covered in ICH Q8(R2). Inclusion of a clear statement of the proposed design space and the location of the filed information (hyperlinked, where possible) in regulatory submissions should be considered to facilitate the regulatory process.
Some aspects of the design space that could be considered for inclusion in the regulatory submission:
• The design space description, including critical and other relevant parameters. The design space can be presented as ranges of material inputs and process parameters, graphical representations, or through more complex mathematical relationships.
• The relationship between the inputs (e.g., material attributes and/or process parameters) and the CQAs, including an understanding of the interactions among the variables.
• Data supporting the design space, such as prior knowledge, conclusions from risk assessments as part of QRM, and experimental studies with supporting data, design assumptions, data analysis, and models.
• The relationship between the proposed design space and other unit operations or process steps.
• Results and conclusions of the studies, if any, of a design space across different scales.
• Justification that the control strategy ensures that the manufacturing process is maintained within the boundaries defined by the design space.
D. Lifecycle Management of a Design Space
The control strategy used for implementation of a design space in production depends on the capabilities of the manufacturing site. The batch records reflect the control strategy used. For example, if a mathematical expression is used for determining a process parameter or a CQA, the batch record would include the input values for variables and the calculated result.
As part of the technology transfer of a design space to a site and throughout the lifecycle, it is important to share the knowledge gained during development and implementation that is relevant for using that design space both on the manufacturing floor and under the PQS of the company or site. This knowledge can include results of risk assessments, assumptions based on prior knowledge, and statistical design considerations. Linkages among the design space, control strategy, CQA, and QTPP are an important part of this shared knowledge.
Each company can decide on the approach used to capture design space information and movements within the design space under the applicable PQS, including additional data gained through manufacturing experience with the design space. In the case of changes to an approved design space, appropriate filings should be made to meet regional regulatory requirements. Movement within the approved design space, as defined in the ICH Q8(R2) glossary, does not call for a regulatory filing. For movement outside the design space, the use of risk assessment could be helpful in determining the impact of the change on quality, safety, and efficacy and the appropriate regulatory filing strategy, in accordance with regional requirements.
VII. WHAT ARE THE POINTS TO CONSIDER FOR PROCESS VALIDATION/CONTINUOUS PROCESS VERIFICATION
These points to consider are intended to illustrate how using principles from ICH Q8(R2), Q9, and Q10 can support an alternative process validation approach and are applicable to drug substance and drug product. They emphasize a more holistic approach to Process Validation across the product lifecycle, including continuous process verification (CPV).
The main objective of process validation is to confirm that a process will consistently yield a product meeting its predefined quality criteria. This can be achieved in different ways, including a traditional approach, CPV, or a combination of these. There are different regional regulatory approaches to process validation. However, the concepts in this document generally are universally accepted, as is the appropriate use of Quality Risk Management (QRM) principles in this context.
In the traditional process validation approach, the focus is on a limited number of batches at discrete time-points during the product lifecycle (e.g., at technology transfer or when changes are introduced). These batches are manufactured at commercial scale using the control strategy with an increased level and frequency of sampling. This validation approach generally remains appropriate, even if enhanced pharmaceutical development has been conducted.
Knowledge gained from development is the foundation for process validation. During technology transfer, site changes, and scale-up, the control strategy can be further developed as new variables are encountered in the commercial manufacturing environment. In many cases, new knowledge will be gained, often leading to modification of the control strategy and improvements to the process, thereby having an impact on process validation. This lifecycle approach to process validation recognizes that elements of process validation
begin with knowledge gained during development, and continues through technology transfer and throughout the commercial manufacturing phase of a product.
A risk-based approach can be used to determine the plan for process validation studies to ensure that process understanding is considered and that the areas of risk are addressed.
B. Continuous Process Verification (CPV)
ICH Q8(R2) describes CPV as an approach to process validation that includes the continuous monitoring and evaluation of manufacturing process performance. Process validation protocols can use CPV for the initial and ongoing commercial production. CPV can also facilitate the evaluation of manufacturing process changes.
CPV can enhance the evaluation of the manufacturing process when it provides substantially more information on process variability and control.
CPV can be applied to an entire process, or to portions of a process, together with traditional process validation approaches.
For initial process validation, CPV generally is more appropriate when an enhanced development approach has been applied. However, it can also be used when extensive process knowledge has been gained through commercial manufacturing experience.
CPV can use in-line, on-line or at-line monitoring or controls to evaluate process performance. These are based on product and process knowledge and understanding. Monitoring can also be combined with feedback loops to adjust the process to maintain output quality. This capability also provides the advantage of enhanced assurance of intra-batch uniformity, fundamental to the objectives of process validation. Some process measurements and controls in support of real-time release testing (RTRT) can also play a role in CPV.
Some advantages of CPV:
• Replaces the emphasis on the first few commercial-scale validation batches with enhanced assurance of product quality in many, or even all, batches;
• Provides the foundation for a robust process performance and product quality monitoring system, increasing product and process knowledge and facilitation of continual improvement opportunities for process and product quality;
• Enables earlier detection of manufacturing-related problems and trends;
• Provides immediate feedback of the effect of a change, thereby facilitating the management of changes;
• Provides a higher assurance of an ongoing state of control, as more data from CPV provide higher statistical confidence for ongoing monitoring and trending;
• Is particularly suited to the evaluation of continuous manufacturing processes; and
• Contributes to the verification of the design space, if used, throughout the product lifecycle.
C. Pharmaceutical Quality System
The PQS strengthens the link between the product lifecycle stages, thereby facilitating the process validation lifecycle approach. Data, information, and knowledge from process performance and product quality monitoring, as described in ICH Q10, support the lifecycle validation approach and the continual improvement of the product and process.
Quality risk management, as an enabler for the PQS, contributes to process validation as follows:
• Risk assessment tools are useful in developing the process validation plan. This can also be useful for the evaluation of the effect of changes.
• Statistical tools support monitoring and trending of process performance to assure a state of control.
Regardless of the approach to process validation, equipment and facilities should be suitably qualified, including computerized systems and control methods, as called for by GMP. Similarly, personnel involved in process validation activities should be appropriately trained and qualified.
[4] Materials for ICH quality training on Q8, Q9, and Q10 are available at www.ich.org under “ICH Training.”
Submit Comments
Submit comments on this guidance document electronically via docket ID: FDA-2013-S-0610 - Specific Electronic Submissions Intended For FDA's Dockets Management Staff (i.e., Citizen Petitions, Draft Proposed Guidance Documents, Variances, and other administrative record submissions)
If unable to submit comments online, please mail written comments to:
Dockets Management
Food and Drug Administration
5630 Fishers Lane, Rm 1061
Rockville, MD 20852
All comments should be identified with the title of the guidance.