BAM Chapter 21A: Examination of Canned Foods
Bacteriological Analytical Manual (BAM) Main Page
January 2001
Authors: Warren L. Landry, Albert H. Schwab, and Gayle A. Lancette
For additional information, contact Steven Simpson
The incidence of spoilage in canned foods is low, but when it occurs it must be investigated properly. Swollen cans often indicate a spoiled product. During spoilage, cans may progress from normal to flipper, to springer, to soft swell, to hard swell. However, spoilage is not the only cause of abnormal cans. Overfilling, buckling, denting, or closing while cool may also be responsible. Microbial spoilage and hydrogen, produced by the interaction of acids in the food product with the metals of the can, are the principal causes of swelling. High summer temperatures and high altitudes may also increase the degree of swelling. Some microorganisms that grow in canned foods, however, do not produce gas and therefore cause no abnormal appearance of the can; nevertheless, they cause spoilage of the product.
Spoilage is usually caused by growth of microorganisms following leakage or underprocessing. Leakage occurs from can defects, punctures, or rough handling. Contaminated cooling water sometimes leaks to the interior through pinholes or poor seams and introduces bacteria that cause spoilage. A viable mixed microflora of bacterial rods and cocci is indicative of leakage, which may usually be confirmed by can examination. Underprocessing may be caused by undercooking; retort operations that are faulty because of inaccurate or improperly functioning thermometers, gauges, or controls; excessive contamination of the product for which normally adequate processes are insufficient; changes in formulation or handling of the product that result in a more viscous product or tighter packing in the container, with consequent lengthening of the heat penetration time; or, sometimes, accidental bypassing of the retort operation altogether. When the can contains a spoiled product and no viable microorganisms, spoilage may have occurred before processing or the microorganisms causing the spoilage may have died during storage.
Underprocessed and leaking cans are of major concern and both pose potential health hazards. However, before a decision can be made regarding the potential health hazard of a low-acid canned food, certain basic information is necessary. Naturally, if Clostridium botulinum (spores, toxin, or both) is found, the hazard is obvious. Intact cans that contain only mesophilic, Gram-positive, sporeforming rods should be considered underprocessed, unless proved otherwise. It must be determined that the can is intact (commercially acceptable seams and no microleaks) and that other factors that may lead to underprocessing, such as drained weight and product formulation, have been evaluated.
The preferred type of tool for can content examination is a bacteriological can opener consisting of a puncturing device at the end of a metal rod mounted with a sliding triangular blade that is held in place by a set screw. The advantage over other types of openers is that it does no damage to the double seam and therefore will not interfere with subsequent seam examination of the can.
Table 1. Useful descriptive terms for canned food analysis.
|
|
||
|
|
|
|
|
|
|
|
| Flat - a can with both ends concave; it remains in this condition even when the can is brought down sharply on its end on a solid, flat surface. | |||
| Flipper - a can that normally appears flat; when brought down sharply on its end on a flat surface, one end flips out. When pressure is applied to this end, it flips in again and the can appears flat. | |||
| Springer - a can with one end permanently bulged. When sufficient pressure is applied to this end, it will flip in, but the other end will flip out. | |||
| Soft swell - a can bulged at both ends, but not so tightly that the ends cannot be pushed in somewhat with thumb pressure. | |||
| Hard swell - a can bulged at both ends, and so tightly that no indentation can be made with thumb pressure. A hard swell will generally "buckle" before the can bursts. Bursting usually occurs at the double seam over the side seam lap, or in the middle of the side seam. | |||
The number of cans examined bacteriologically should be large enough to give reliable results. When the cause of spoilage is clear-cut, culturing 4-6 cans may be adequate, but in some cases it may be necessary to culture 10-50 cans before the cause of spoilage can be determined. On special occasions these procedures may not yield all the required information, and additional tests must be devised to collect the necessary data. Unspoiled cans may be examined bacteriologically to determine the presence of viable but dormant organisms. The procedure is the same as that used for spoiled foods except that the number of cans examined and the quantity of material subcultured must be increased.
A. Equipment and materials
- Incubators, thermostatically controlled at 30, 35, and 55°C
- pH meter, potentiometer
- Microscope, slides, and coverslips
- Can opener, bacteriological can opener, and can punch, all sterile
- Petri dishes, sterile
- Test tubes, sterile
- Serological pipets, cotton-plugged, sterile
- Nontapered pipets, cotton-plugged (8 mm tubing), sterile
- Soap, water, brush, and towels, sterile and nonsterile
- Indelible ink marking pen
- Diamond point pen for marking cans
- Examination pans (Pyrex or enamel baking pans)
- Bromcresol purple (BCP) dextrose broth (M27)
- Chopped liver broth (M38) or cooked meat medium (CMM) (M42)
- Malt extract broth (M94)
- Liver-veal agar (without egg yolk) (LVA) (M83)
- Acid broth (M4)
- Nutrient agar (NA) (M112)
- Methylene blue stain (R45), crystal violet (R16), or Gram stain (R32)
- Sabouraud's dextrose agar (SAB) (M133)
- 4% Iodine in 70% ethanol (R18)
C. Can preparation
Remove labels. With marking pen, transfer subnumbers to side of can to aid in correlating findings with code. Mark labels so that they may be replaced in their original position on the can to help locate defects indicated by stains on label. Separate all cans by code numbers and record size of container, code, product, condition, evidence of leakage, pinholes or rusting, dents, buckling or other abnormality, and all identifying marks on label. Classify each can according to the descriptive terms in Table 1. Before observing cans for classification, make sure cans are at room temperature.
D. Examination of can and contents
Classification of cans. NOTE: Cans must be at room temperature for classification.
- Sampling can contents
-
Swollen cans. Immediately analyze springers, swells, and a representative number (at least 6, if available) of flat and flipper cans. Retain examples of each, if available, when reserve portion must be held. Place remaining flat and flipper cans (excluding those held in reserve) in incubator at 35°C. Examine at frequent intervals for 14 days. When abnormal can or one becoming increasingly swollen is found, make note of it. When can becomes a hard swell or when swelling no longer progresses, culture sampled contents, examine for preformed toxin of C. botulinum if microscopic examination shows typical C. botulinum organisms or Gram-positive rods, and perform remaining steps of canned food examination.
- Flat and flipper cans. Place cans (excluding those held in reserve) in incubator at 35°C. Observe cans for progressive swelling at frequent intervals for 14 days. When swelling occurs, follow directions in l-a, above. After 14 days remove flat and flipper cans from incubator and test at least 6, if available. (It is not necessary to analyze all normal cans.) Do not incubate cans at temperatures above 35°C. After incubation, bring cans back to room temperature before classifying them.
-
- Opening the can. Open can in an environment that is as aseptic as possible. Use of vertical laminar flow hood is recommended.
-
Hard swells, soft swells, and springers. Chill hard swells in refrigerator before opening. Scrub entire uncoded end and adjacent sides of can using abrasive cleanser, cold water, and a brush, steel wool, or abrasive pad. Rinse and dry with clean sterile towel. Sanitize can end to be opened with 4% iodine in 70% ethanol for 30 min and wipe off with sterile towel. DO NOT FLAME. Badly swollen cans may spray out a portion of the contents, which may be toxic. Take some precaution to guard against this hazard, e.g., cover can with sterile towel or invert sterile funnel over can. Sterilize can opener by flaming until it is almost red, or use separate presterilized can openers, one for each can. At the time a swollen can is punctured, test for headspace gas, using a qualitative test or the gas-liquid chromatography method described below. For a qualitative test, hold mouth of sterile test tube at puncture site to capture some escaping gas, or use can-puncturing press to capture some escaping gas in a syringe. Flip mouth of tube to flame of Bunsen burner. A slight explosion indicates presence of hydrogen. Immediately turn tube upright and pour in a small amount of lime water. A white precipitate indicates presence of CO2. Make opening in sterilized end of can large enough to permit removal of sample.
- Flipper and flat cans. Scrub entire uncoded end and adjacent sides of can using abrasive cleanser, warm water, and a brush, steel wool, or abrasive pad. Rinse and dry with clean sterile towel. Gently shake cans to mix contents before sanitizing. Flood end of can with iodine-ethanol solution and let stand at least 15 min. Wipe off iodine mixture with clean sterile towel. Ensure sterility of can end by flaming with burner in a hood until iodine-ethanol solution is burned off, end of can becomes discolored from flame, and heat causes metal to expand. Be careful not to inhale iodine fumes while burning off can end. Sterilize can opener by flaming until it is almost red, or use separate presterilized can openers for each can. Make opening in sterilized end of can large enough to permit removal of sample.
-
-
Removal of material for testing. Remove large enough portions from center of can to inoculate required culture media. Use sterile pipets, either regular or wide-mouthed. Transfer solid pieces with sterile spatulas or other sterile devices. Always use safety devices for pipetting. After removal of inocula, aseptically transfer at least 30 ml or, if less is available, all remaining contents of cans to sterile closed containers, and refrigerate at about 4°C. Use this material for repeat examination if needed and for possible toxicity tests. This is the reserve sample. Unless circumstances dictate otherwise, analyze normal cans submitted with sample organoleptically and physically (see 5-b, below), including pH determination and seam teardown and evaluation. Simply and completely describe product appearance, consistency, and odor on worksheet. If analyst is not familiar with decomposition odors of canned food, another analyst, preferably one familiar with decomposition odors, should confirm this organoleptic evaluation. In describing the product in the can, include such things as low liquid level (state how low), evidence of compaction, if apparent, and any other characteristics that do not appear normal. Describe internal and external condition of can, including evidence of leakage, etching, corrosion, etc.
-
Physical examination. Perform net weight determinations on a representative number of cans examined (normal and abnormal). Determine drained weight, vacuum, and headspace on a representative number of normal-appearing and abnormal cans (1). Examine metal container integrity of a representative number of normal cans and all abnormal cans that are not too badly buckled for this purpose (see Chapter 22). CAUTION: Always use care when handling the product, even apparently normal cans, because botulinal toxin may be present.
- Cultural examination of low-acid food (pH greater than 4.6). If there is any question as to product pH range, determine pH of a representative number of normal cans before proceeding. From each container, inoculate 4 tubes of chopped liver broth or cooked meat medium previously heated to 100°C (boiling) and rapidly cooled to room temperature; also inoculate 4 tubes of bromcresol purple dextrose broth. Inoculate each tube with 1-2 ml of product liquid or product-water mixture, or 1-2 g of solid material. Incubate as in Table 2.
Table 2. Incubation times for various media for examination of low acid foods (pH > 4.6).Medium No. of tubes Temp. (°C) Time of incubation (h) Chopped liver (cooked meat) 2 35 96-120 Chopped liver (cooked meat) 2 55 24-72 Bromcresol purple dextrose broth 2 55 24-48 Bromcresol purple dextrose broth 2 35 96-120
After culturing and removing reserve sample, test material from cans (other than those classified as flat) for preformed toxins of C. botulinum when appropriate, as described in Chapter 17.
-
Microscopic examination. Prepare direct smears from contents of each can after culturing. Dry, fix, and stain with methylene blue, crystal violet, or Gram stain. If product is oily, add xylene to a warm, fixed film, using a dropper; rinse and stain. If product washes off slide during preparation, examine contents as wet mount or hanging drop, or prepare suspension of test material in drop of chopped liver broth before drying. Check liver broth before use to be sure no bacteria are present to contribute to the smear. Examine under microscope; record types of bacteria seen and estimate total number per field.
- Physical and organoleptic examination of can contents. After removing reserve sample from can, determine pH of remainder, using pH meter. DO NOT USE pH PAPER. Pour contents of cans into examination pans. Examine for odor, color, consistency, texture, and overall quality. DO NOT TASTE THE PRODUCT. Examine can lining for blackening, detinning, and pitting.
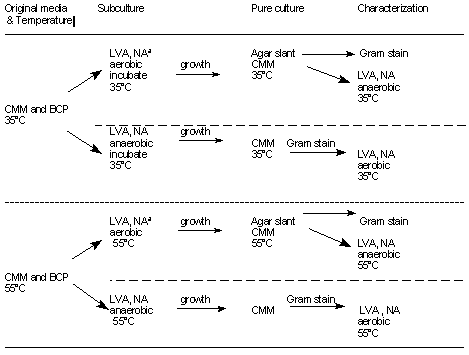
Table 3. Schematic diagram of culture procedure for low-acid canned foods
a LVA, liver-veal agar; NA, nutrient agar; CMM, cooked meat medium; BCP, bromcresol purple dextrose broth.
Table 4. Incubation of acid broth and malt extract broth used for acid foods (pH 4.6)
| Medium | No. of tubes | Temp. (°C) | Time of incubation (h) |
|---|---|---|---|
| Acid broth | 2 | 55 | 48 |
| Acid broth | 2 | 30 | 96 |
| Malt extract broth | 2 | 30 | 96 |
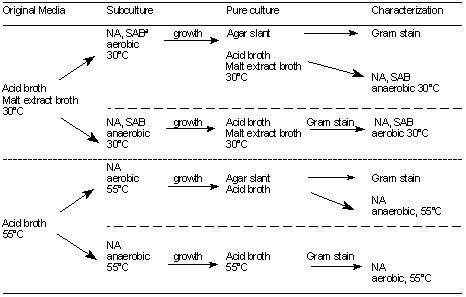
Table 5. Pure culture scheme for acid foods (pH 4.6).
a NA, nutrient agar; SAB, Sabouraud's dextrose agar.
E. Cultural findings in cooked meat medium (CMM) and bromcresol purple dextrose broth (BCP)
Check incubated medium for growth at frequent intervals up to maximum time of incubation (Table 2). If there is no growth in either medium, report and discard. At time growth is noted streak 2 plates of liver-veal agar (without egg yolk) or nutrient agar from each positive tube. Incubate one plate aerobically and one anaerobically, as in schematic diagram (Table 3). Reincubate CMM at 35°C for maximum of 5 days for use in future toxin studies. Pick representatives of all morphologically different types of colonies into CMM and incubate for appropriate time, i.e., when growth is sufficient for subculture. Dispel oxygen from CMM broths to be used for anaerobes but not from those to be used for aerobes. After obtaining pure isolates, store cultures to maintain viability.
- If mixed microflora is found only in BCP, report morphological types. If rods are included among mixed microflora in CMM, test CMM for toxin, as described in Chapter 17. If Gram-positive or Gram-variable rods typical of either Bacillus or Clostridium organisms are found in the absence of other morphological types, search to determine whether spores are present. In some cases, old vegetative cells may appear to be Gram-negative and should be treated as if they are Gram-positive. Test culture for toxin according to Chapter 17.
-
Table 6. Classification of food products according to acidity Low acid--pH greater than 4.6 Acid pH 4.6 and below Meats Tomatoes Seafoods Pears Milk Pineapple - Meat and vegetable Mixtures and "specialties"
- Spaghetti
- Soups
Other fruit Sauerkraut - Vegetables
- Asparagus
- Beets
- Pumpkin
- Green beans
- Corn
- Lima beans
Pickles Berries Citrus Rhubarb Table 7. Spoilage microorganisms that cause high and low acidity in various vegetables and fruits Spoilage type pH groups Examples Thermophilic Flat-sour >5.3 Corn, peas Thermophilic(a) >4.8 Spinach, corn Sulfide spoilage(a) >5.3 Corn, peas Mesophilic Putrefactive anaerobes(a) >4.8 Corn, asparagus Butyric anaerobes >4.0 Tomatoes, peas Aciduric flat-sour(a) >4.2 Tomato juice Lactobacilli 4.5-3.7 Fruits Yeasts 3.7 Fruits Molds 3.7 Fruits a The responsible organisms are bacterial sporeformers. Table 8. Spoilage manifestations in low-acid products Group of organisms Classification Manifestations Flat-sour Can flat Possible loss of vacuum on storage Product Appearance not usually altered; pH markedly lowered, sour; may have slightly abnormal odor; sometimes cloudy liquor Thermophilic anaerobe Can swells May burst Product Fermented, sour, cheesy or butyric odor Sulfide spoilage Can flat H2S gas absorbed by product Product Usually blackened; rotten egg odor Putrefactive anaerobe Can swells May burst Product May be partially digested; pH slightly above normal; typical putrid odor Aerobic sporeformers Can flat or swollen Usually no swelling, except in cured meats when nitrate and sugar present; coagulated evaporated milk, black beets Table 9. Spoilage manifestations in acid products Type of organism Classification Manifestation Bacillus thermoacidurans (flat, sour tomato juice) Can flat Little change in vacuum Product Slight pH change; off-odor Butyric anaerobes (tomatoes and tomato juice) Can swells May burst Product Fermented, butyric odor Nonsporeformers (mostly lactic types) Can swells Usually burst, but swelling may be arrested Product Acid odor Table 10. Laboratory diagnosis of bacterial spoilage Underprocessed Leakage Can Flat or swelled; seams generally normal Swelled; may show normal defects(a) Product appearance Sloppy or fermented Frothy fermentation; viscous odor Normal, sour or putrid, but generally consistent from can to can Sour, fecal; generally varying from can to can pH Usually fairly constant Wide variation Microscopic and cultural Cultures show sporeforming rods only Mixed cultures, generally rods and cocci; only at usual temperatures Growth at 35 and/or 55°C. May be characteristic on special growth media, e.g., acid agar for tomato juice.
If product misses retort completely, rods, cocci,yeast or molds, or any combination of these may be present.
History Spoilage usually confined to certain portions of pack Spoilage scattered In acid products, diagnosis may be less clearly defined; similar organisms may be involved in understerilization and leakage. a Leakage may be due not to can defects but to other factors, such as contamination of cooling water or rough handling, e.g., can unscramblers, rough conveyor system. Table 11. pH range of a few selected commercially canned foods Food pH range Food pH range Apples, juice 3.3 - 3.5 Jam, fruit 3.5 - 4.0 Apples, whole 3.4 - 3.5 Jellies, fruit 3.0 - 3.5 Asparagus, green 5.0 - 5.8 Lemon juice 2.2 - 2.6 Beans Lemons 2.2 - 2.4 Baked 4.8 - 5.5 Lime juice 2.2 - 2.4 Green 4.9 - 5.5 Loganberries 2.7 - 3.5 Lima 5.4 - 6.3 Mackerel 5.9 - 6.2 Soy 6.0 - 6.6 Milk Beans with pork 5.1 - 5.8 Cow, whole 6.4 - 6.8 Beef, corned, hash 5.5 - 6.0 Evaporated 5.9 - 6.3 Beets, whole 4.9 - 5.8 Molasses 5.0 - 5.4 Blackberries 3.0 - 4.2 Mushroom 6.0 - 6.5 Blueberries 3.2 - 3.6 Olives, ripe 5.9 - 7.3 Boysenberries 3.0 - 3.3 Orange juice 3.0 - 4.0 Bread Oysters 6.3 - 6.7 White 5.0 - 6.0 Peaches 3.4 - 4.2 Date and nut 5.1 - 5.6 Pears (Bartlett) 3.8 - 4.6 Broccoli 5.2 - 6.0 Peas 5.6 - 6.5 Carrot juice 5.2 - 5.8 Pickles Carrots, chopped 5.3 - 5.6 Dill 2.6 - 3.8 Cheese Sour 3.0 - 3.5 Parmesan 5.2 - 5.3 Sweet 2.5 - 3.0 Roquefort 4.7 - 4.8 Pimento 4.3 - 4.9 Cherry juice 3.4 - 3.6 Pineapple Chicken 6.2 - 6.4 Crushed 3.2 - 4.0 Chicken with noodles 6.2 - 6.7 Juice 3.4 - 3.7 Chop suey 5.4 - 5.6 Sliced 3.5 - 4.1 Cider 2.9 - 3.3 Plums 2.8 - 3.0 Clams 5.9 - 7.1 Potato salad 3.9 - 4.6 Cod fish 6.0 - 6.1 Potatoes Corn Mashed 5.1 Cream style 5.9 - 6.5 White, whole 5.4 - 5.9 On-the-cob 6.1 - 6.8 Prune juice 3.7 - 4.3 Whole grain Pumpkin 5.2 - 5.5 Brine-packed Raspberries 2.9 - 3.7 Vacuum-packed 6.0 - 6.4 Rhubarb 2.9 - 3.3 Crab apples, spiced 3.3 - 3.7 Salmon 6.1 - 6.5 Cranberry Sardines 5.7 - 6.6 Juice 2.5 - 2.7 Sauerkraut 3.1 - 3.7 Sauce 2.3 Juice 3.3 - 3.4 Currant juice 3.0 Shrimp 6.8 - 7.0 Dates 6.2 - 6.4 Soups Duck 6.0 - 6.1 Bean 5.7 - 5.8 Figs 4.9 - 5.0 Beef broth 6.0 - 6.2 Frankfurters 6.2 - 6.2 Chicken noodle 5.5 - 6.5 Fruit cocktail 3.6 - 4.0 Clam chowder 5.6 - 5.9 Gooseberries 2.8 - 3.1 Duck 5.0 - 5.7 Grapefruit Mushroom 6.3 - 6.7 Juice 2.9 - 3.4 Noodle 5.6 - 5.8 Pulp 3.4 Oyster 6.5 - 6.9 Sections 3.0 - 3.5 Pea 5.7 - 6.2 Grapes 3.5 - 4.5 Spinach 4.8 - 5.8 Ham, spiced 6.0 - 6.3 Squash 5.0 - 5.8 Hominy, lye 6.9 - 7.9 Tomato 4.2 - 5.2 Huckleberries 2.8 - 2.9 Turtle 5.2 - 5.3 Vegetable 4.7 - 5.6 Strawberries 3.0 - 3.9 Miscellaneous products Sweet potatoes 5.3 - 5.6 Beers 4.0 - 5.0 Tomato juice 3.9 - 4.4 Ginger ale 2.0 - 4.0 Tomatoes 4.1 - 4.4 Human Tuna 5.9 - 6.1 Blood plasma 7.3 - 7.5 Turnip greens 5.4 - 5.6 Duodenal contents 4.8 - 8.2 Vegetable juice 3.9 - 4.3 Feces 4.6 - 8.4 Vegetables, mixed 5.4 - 5.6 Gastric contents 1.0 - 3.0 Vinegar 2.4 - 3.4 Milk 6.6 - 7.6 Youngberries 3.0 - 3.7 Saliva 6.0 - 7.6 Spinal fluid 7.3 - 7.5 Urine 4.8 - 8.4 Magnesia, milk of 10.0 -10.5 Water Distilled, CO2 6.8 - 7.0 Mineral 6.2 - 9.4 Sea 8.0 - 8.4 Wine 2.3 - 3.8 - If no toxin is present, send pure cultures for evaluation of heat resistance to Cincinnati District Office, FDA, 1141 Central Parkway, Cincinnati, OH 45202, if cultures meet the following criteria:
-
Cultures come from intact cans that are free of leaks and have commercially acceptable seams. (Can seams of both ends of can must be measured; visual examination alone is not sufficient.)
- Two or more tubes are positive and contain similar morphological types.
-
- Examination of acid foods (pH 4.6 and below) by cultivation. From each can, inoculate 4 tubes of acid broth and 2 tubes of malt extract broth with 1-2 ml or 1-2 g of product, using the same procedures as for low-acid foods, and incubate as in Table 4. Record presence or absence of growth in each tube, and from those that show evidence of growth, make smears and stain. Report types of organisms seen. Pure cultures may be isolated as shown in Table 5.
G. Interpretation of results (see Tables 6-11)
-
The presence of only sporeforming bacteria, which grow at 35°C, in cans with satisfactory seams and no microleaks indicates underprocessing if their heat resistance is equal to or less than that of C. botulinum. Spoilage by thermophilic anaerobes such as C. thermobutylicum may be indicated by gas in cooked meat at 55°C and a cheesy odor. Spoilage by C. botulinum, C. sporogenes, or C. perfringens may be indicated in cooked meat at 35°C by gas and a putrid odor; rods, spores, and clostridial forms may be seen on microscopic examination. Always test supernatants of such cultures for botulinal toxin even if no toxin was found in the product itself, since viable botulinal spores in canned foods indicate a potential public health hazard, requiring recall of all cans bearing the same code. Spoilage by mesophilic organisms such as Bacillus thermoacidurans or B. coagulans and/or thermophilic organisms such as B. stearothermophilus, which are flat-sour types, may be indicated by acid production in BCP tubes at 35 and/or 55°C in high-acid or low-acid canned foods. No definitive conclusions may be drawn from inspection of cultures in broth if the food produced an initial turbidity on inoculation. Presence or absence of growth in this case must be determined by subculturing.
-
Spoilage in acid products is usually caused by nonsporeforming lactobacilli and yeasts. Cans of spoiled tomatoes and tomato juice remain flat but the products have an off-odor, with or without lowered pH, due to aerobic, mesophilic, and thermophilic sporeformers. Spoilage of this type is an exception to the general rule that products below pH 4.6 are immune to spoilage by sporeformers. Many canned foods contain thermophiles which do not grow under normal storage conditions, but which grow and cause spoilage when the product is subjected to elevated temperatures (50-55°C). B. thermoacidurans and B. stearothermophilus are thermophiles responsible for flat-sour decomposition in acid and low-acid foods, respectively. Incubation at 55°C will not cause a change in the appearance of the can, but the product has an off-odor with or without a lowered pH. Spoilage encountered in products such as tomatoes, pears, figs, and pineapples is occasionally caused by C. pasteurianum, a sporeforming anaerobe which produces gas and a butyric acid odor. C. thermosaccolyticum is a thermophilic anaerobe which causes swelling of the can and a cheesy odor of the product. Cans which bypass the retort without heat processing usually are contaminated with nonsporeformers as well as sporeformers, a spoilage characteristic similar to that resulting from leakage.
-
A mixed microflora of viable bacterial rods and cocci usually indicates leakage. Can examination may not substantiate the bacteriological findings, but leakage at some time in the past must be presumed. Alternatively, the cans may have missed the retort altogether, in which case a high rate of swells would also be expected.
-
A mixed microflora in the product, as shown by direct smear, in which there are large numbers of bacteria visible but no growth in the cultures, may indicate precanning spoilage. This results from bacterial growth in the product before canning. The product may be abnormal in pH, odor, and appearance.
-
If no evidence of microbial growth can be found in swelled cans, the swelling may be due to development of hydrogen by chemical action of contents on container interiors. The proportion of hydrogen varies with the length and condition of storage. Thermophilic anaerobes produce gas, and since cells disintegrate rapidly after growth, it is possible to confuse thermophilic spoilage with hydrogen swells. Chemical breakdown of the product may result in evolution of carbon dioxide. This is particularly true of concentrated products containing sugar and some acid, such as tomato paste, molasses, mincemeats, and highly sugared fruits. The reaction is accelerated at elevated temperatures.
- Any organisms isolated from normal cans that have obvious vacuum and normal product but no organisms in the direct smear should be suspected as being a laboratory contaminant. To confirm, aseptically inoculate growing organism into another normal can, solder the hole closed, and incubate 14 days at 35°C. If any swelling of container or product changes occur, the organism was probably not in the original sample. If can remains flat, open it aseptically and subculture as previously described. If a culture of the same organism is recovered and the product is normal, consider the product commercially sterile since the organism does not grow under normal conditions of storage and distribution.
Headspace Gas Determination by Gas-Liquid Chromatography
Nitrogen, the principal gas normally present in canned foods during storage, is associated with lesser quantities of carbon dioxide and hydrogen. Oxygen included in the container at the time of closure is initially dissipated by container corrosion and/or product oxidation. Departure from this normal pattern can serve as an important indication of changes within the container, since the composition of headspace gases may distinguish whether bacterial spoilage, container corrosion, or product deterioration is the cause of swollen cans (2). Use of the gas chromatograph for analyzing headspace gases of abnormal canned foods has eliminated the possibility of false-negative tests for different gases. It has also allowed the analyst to determine the percentage of each gas present, no matter what the mixture is. By knowing these percentages, the analyst can be alerted to possible can deterioration problems or bacterial spoilage. A rapid gas-liquid chromatographic procedure is presented here for the determination of carbon dioxide, hydrogen, oxygen, nitrogen, and hydrogen sulfide from the headspace of abnormal canned foods.
The analysis of 2352 abnormal canned foods, composed of 288 different products by a gas-liquid chromatography showed viable microorganisms in 256 cans (3). Analysis of this data showed that greater than 10 percent carbon dioxide in the headspace gas was indicative of microbial growth. Although greater than 10 percent carbon dioxide is found in a container, long periods of storage at normal temperatures can result in autosterilization and absence of viable microorganisms. Carbon dioxide my be produced in sufficient quantities to swell the container. Storage at elevated temperatures accelerates this action. Hydrogen can be produced in cans when the food contents react chemically with the metal of the seam (3).
A. Equipment and materials
-
Fisher Model 1200 Gas Partitioner, with dual thermal conductivity cells and dual in-line columns. Column No. 1 is 6-1/2 ft × 1/8 inch, aluminum packed, with 80-100 mesh ColumpakTM PQ. Column No. 2 is 11 ft × 3/16 inch, aluminum packed, with 60-80 mesh molecular sieve 13× (Fig. 1).
NOTE: Other gas chromatograph instruments equipped with the appropriate columns, carrier gas, detector and recorder or integrator may also be suitable for this analysis.
Operating conditions: column temperature, 75°C; attenuation, 64/256; carrier gas, argon, with in-let pressure of 40 psig; flow rate, 26 ml/min through gas partitioner and 5 ml/min through flush line; bridge current, 125 mA; column mode, 1 & 2; temperature mode, column; injector temperature, off.
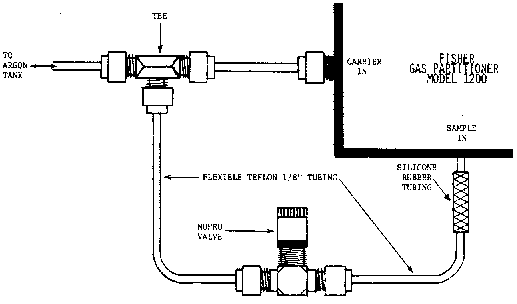
NOTE: Installation of flush system. Injection of gas samples through either sample out port or septum injection port may lead to damaged filaments in detector and excessive accumulation of moisture on columns due to bypassing the sample drying tube. To avoid this, make all injections in the sample in port. To avoid cross-contamination, install a flush line off the main argon line (Fig. 2), and flush sample loop between injections.
-
Strip chart recorder, with full scale deflection and speed set at 1 cm/min, 1 mv
-
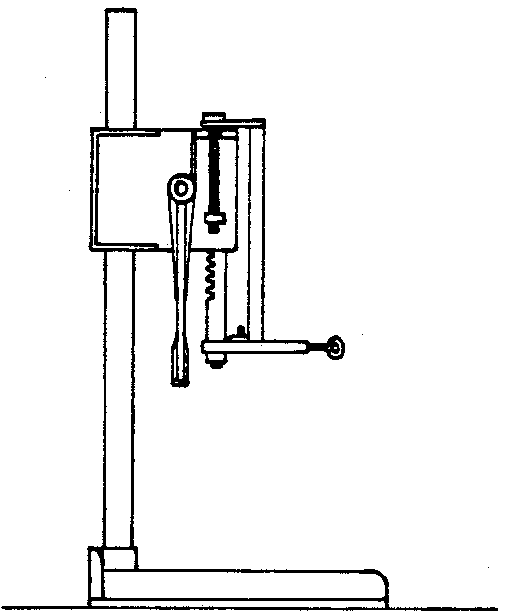
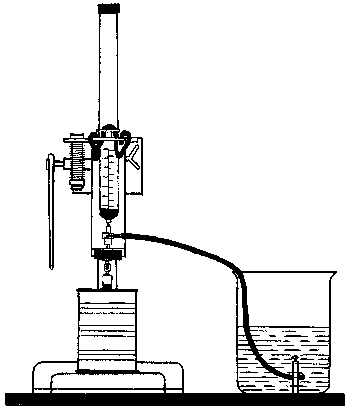
Can puncturing press (Fig. 3)
-
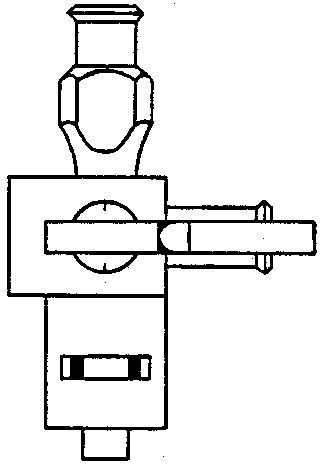
Sterile stainless steel gas piercers (Fig. 4)
-
Miniature inert valve, with 3-way stopcock and female luer on left side (Popper & Sons, Inc., 300 Denton Ave., New Hyde Park, NY 11040), or equivalent (Fig. 5)
-
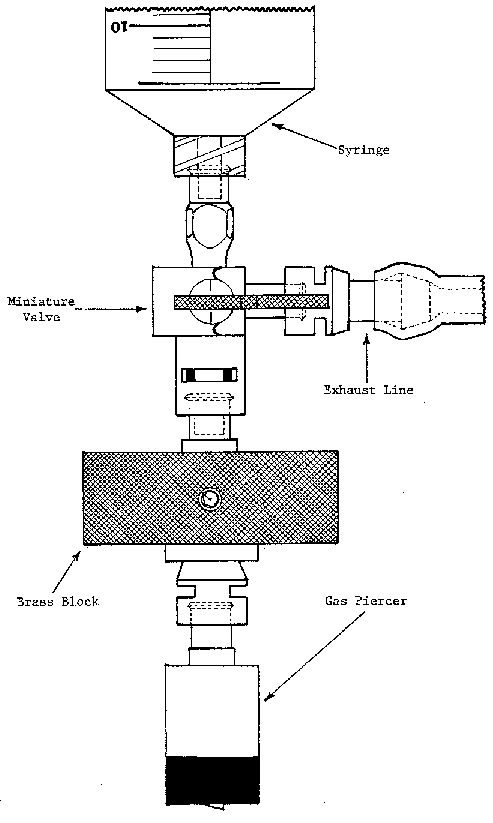
Plastic disposable 10-50 ml syringes, with restraining attachment for maximum volume control (Fig. 6). Syringes may be reused.
-
Gas chromatograph and caps, for capping syringes (Alltech Associates, Inc., 202 Campus Drive, Arlington Heights, IL 60004), or equivalent (Fig. 6)
-
Beaker, 1 liter, glass or metal
-
Plastic gas tubing, 3 ft × 1/8 inch id, for exhaust tubing
-
Soap solution, for detecting gas leaks ("SNOOP" Nuclear Products Co., 15635 Saranac Road, Cleveland, OH 44110), or equivalent
-
Small pinch clamp, to weigh down exhaust tubing in beaker
-
Nupro Valve, flow-regulating valve for flush line, 1/8 inch, Angle Pattern Brass (Alltech), or equivalent (Fig. 2)
- Silicone rubber tubing, seamless, red, autoclavable, 1/8 inch bore × 3/16 inch wall thickness (Arthur H. Thomas Co., Vine St. at 3rd, Philadelphia, PA), or equivalent
B. Calibration of gas chromatograph
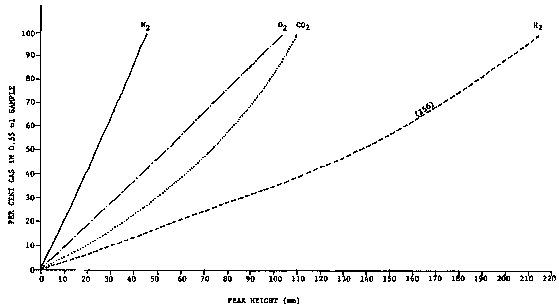
Calibration gases of known proportions are commercially available. Construct calibration curves from analysis of pure gases and at least 2-3 different percentage mixtures of gases. Plot linear graph of various known concentrations of each gas as peak height (mm) vs percent gas (Fig. 7).
C. Preparation of materials
Prepare gas collection apparatus as illustrated in Figs. 8 and 9. Adjust height of gas collection apparatus to height of can to be examined. Attach male terminal of miniature valve to female Luer-Lok terminal mounted on top of brass block on can-puncturing press. Attach one end of gas exhaust tubing to female terminal of miniature valve. Attach small pinch clamp to other end of gas exhaust tubing and place in beaker partially filled with water. Attach disposable syringe to other female Luer-Lok terminal on miniature valve. Turn 2-way plug so that gas entering from piercer will flow toward disposable syringe. Place sterile gas piercer in position on male terminal mounted on bottom of brass block on can-puncturing press.
D. Collection of headspace gas
Place can under gas press (cans to be cultured should first be cleaned and sterilized). Lower handle until gas piercer punctures can and seals. Hold in position until adequate volume of gas has been collected (minimum of 5 ml); then turn 2-way plug to release excess gas through exhaust tubing. Release handle, remove syringe, and cap immediately. Identify syringe appropriately.
E. Injection of gas into gas chromatograph
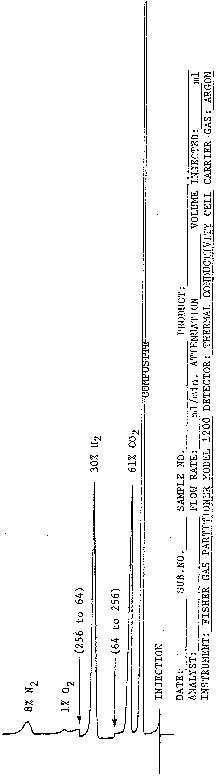
Turn on gas chromatograph and recorder. Let stabilize for about 2 h. Make sure flush line is attached and gas sampling valve is open to allow flushing of sample loop. Turn on chart drive on recorder. Remove flush line, uncap, and immediately attach syringe to Sample-In Injection Port. Inject 5-10 ml of gas and immediately close gas sampling valve. Remove syringe and cap. Reattach flush line onto Sample-In Port and open gas sample valve to allow flushing of system before next injection. Observe chromatogram and switch attenuation from 64 to 256 after carbon dioxide peak has been recorded and returned back to base line. This allows hydrogen peak to be retained on scale. After hydrogen peak returns to base line, switch attenuation back to 64. After instrument has separated gases (about 6 min), determine retention time and peak height for each gas recovered from unknown sample and percent determined from standard graph by comparing retention times and peak heights with known gases, usually associated with headspace gases from abnormal canned food products. Mount chromatogram on mounting paper and identify properly as in Fig. 10. For each sample examined, inject control gases for each type of headspace gas recovered.
Figure 2. Flush System.
Figure 3. Can puncturing press.
Figure 6. Plastic disposable syringe with restraining attachment.
Figure 10. Gas chromatograph of headspace gas.
References
- Association of Official Analytical Chemists. 1990. Official Methods of Analysis, 15th ed. AOAC, Arlington, VA.
- Vosti, D.C., H.H. Hernandez, and J.G. Strand. 1961. Analysis of headspace gases in canned foods by gas chromatography. Food Technol. 15:29-31.
- Landry, W.I., J.E. Gilchrist, S. McLaughlin, and J.T. Peeler 1988. Analysis of abnormal canned foods. AOAC Abstracts.
Hypertext Source: Examination of Canned Foods, Bacteriological Analytical Manual, 8th Edition, Revision A, 1998. Chapter 21A.