Unomedical A/S Recalls VariSoft Infusion Sets Due to Damage to the Connector Piece Causing Unexpected Disconnections
The FDA has identified this as a Class I recall, the most serious type of recall. Use of these devices may cause serious injuries or death.
Recalled Product
- Product Names: VariSoft Infusion Sets
- Product Codes: FPA
- Model Numbers: 1002827, 1002828, 1002830
- Lot Numbers: 5388367; 5388357; 5388371; 5388362; 5388368; 5388366; 5388372; 5388376
- Manufacturing Dates: April 1, 2022 to August 1, 2022
- Distribution Dates: October 25, 2022 to September 15, 2023
- Devices Recalled in the U.S.: 13,069 boxes, 10 sets per box
- Date Initiated by Firm: October 11, 2023
Device Use
VariSoft is an infusion set used with Tandem insulin pumps for the subcutaneous infusion of insulin in the treatment of diabetes. With an insertion angle between 20-45°, the infusion set allows users to choose their most comfortable insertion angle. It is often preferred by users who are thin or who have scar tissue or limiting potential insertion sites. This product is delivered sterile and ready for use.
Reason for Recall
Unomedical A/S is recalling certain VariSoft Infusion Sets because there is a risk the connector may detach from the infusion set which will disrupt the delivery of the insulin to the patient. If this occurs, insulin may continue to flow through the tubing that is now disconnected from the patient and the insulin pump would not recognize an infusion set problem and would not alert users.
The defect reported was caused by damage to the connector piece during manufacturing, resulting in the connector becoming detached more easily from the insulin set than expected. Disconnections occurring during sleep, or when otherwise not detected, may lead to hyperglycemia unless it is noticed and reconnected promptly.
There is a chance of death or life-threatening harm to individual patients using devices from these lots, requiring professional medical intervention.
There has been 1 reported injury. There have been no reports of death.
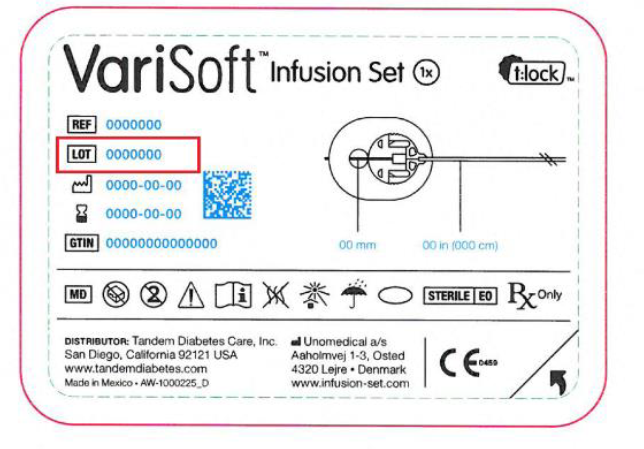
The following lot numbers are in the scope of this recall: 5388367; 5388357; 5388371; 5388362; 5388368; 5388366; 5388372; 5388376
The Lot number is located next to the symbol on the box and pouch labels:
Who May be Affected
- All users of the affected VariSoft Infusion Sets in conjunction with the Tandem insulin pump.
- Children and elderly who may not notice if the connector is detached or may not be able to communicate the disconnection or symptoms of hyperglycemia.
- Children or active sleepers may be at higher risk for disconnection during sleep, when it is less likely to be noticed.
- Pregnant women and their developing fetus.
What to Do
On October 11, 2023, Unomedical A/S sent Tandem Diabetes Care, their sole consignee, an Urgent Field Safety Notice. Tandem Diabetes Care began notifying their customers on October 25, 2023.
The notice requested customers to:
- Not use any VariSoft Infusion Sets with the affected lot numbers in your possession and let Tandem Customer Technical Support know by sending an email or calling.
- Tandem will facilitate the return of the affected product and ship replacement products.
- If all remaining stock is from the affected lots, please immediately contact your Healthcare Provider for guidance and instant replenishment.
- Acknowledge receipt of the letter by sending an email to Tandem Customer Technical Support at Techsupport@tandemdiabetes.com or calling 1-877-801-6901.
Contact Information
Customers in the U.S. with questions about this recall should contact Tandem Customer Technical Support at 1-877-801-6901 or Techsupport@tandemdiabetes.com.
Additional Resources:
- Medical Device Recall Database Entry - Model Number 1002827
- Medical Device Recall Database Entry – Model Number 1002828
- Medical Device Recall Database Entry – Model Number 1002830
How do I report a problem?
Health care professionals and consumers may report adverse reactions or quality problems they experienced using these devices to MedWatch: The FDA Safety Information and Adverse Event Reporting Program using an online form, regular mail, or FAX.