Drug Trials Snapshots: JEUVEAU
HOW TO USE THIS SNAPSHOT
The information provided in Snapshots highlights who participated in the clinical trials that supported the FDA approval of this drug, and whether there were differences among sex, race and age groups. The “MORE INFO” bar shows more detailed, technical content for each section. The Snapshot is intended as one tool for consumers to use when discussing the risks and benefits of the drugs.
LIMITATIONS OF THIS SNAPSHOT
Do not rely on Snapshots to make decisions regarding medical care. Always speak to your health provider about the risks and benefits of a drug. Refer to the JEUVEAU Package Insert for complete information.
JEUVEAU (botulinum toxin- type A)
ju vo
Evolus
Approval date: February 1, 2019
DRUG TRIALS SNAPSHOT SUMMARY:
What is the drug for?
JEUVEAU is a drug used in adults to temporarily improve the appearance of moderate to severe glabellar lines (wrinkles between the eyebrows).
How is this drug used?
JEUVEAU is given by a healthcare provider directly into the muscle through a needle (known as an intramuscular injection). It is injected into five different sites of the muscles of the frown lines. JEUVEAU should not be given more frequently than every three months.
What are the benefits of this drug?
In two trials, patients with moderate to severe wrinkles between the eyebrows, had improvement of frown lines thirty days after treatment with JEUVEAU. In Trial 1, 67% of patients had temporary improvement of wrinkles between the eyebrows compared to 1% of patients in the group that received placebo. In Trial 2, 71% of patients had temporary improvement of wrinkles between the eyebrows compared to 1% of patients in the group that received placebo.
What are the benefits of this drug (results of trials used to assess efficacy)?
The table below summarizes efficacy results for the evaluated patients in the clinical trials. The primary endpoint was the proportion of patients achieving ≥2-grade improvement from baseline at maximum frown, as assessed independently by both the investigator and the patient using the Glabellar Line Scale (GLS) at Day 30.
Table 2. Trials 1 and 2: Composite Investigator and Patient Assessment of Glabellar Line Severity at Maximum Frown at Day 30 – Responder Rates (% of Patients Achieving ≥ 2-Grade Improvement from Baseline)
|
Trial |
JEUVEAU |
Placebo |
|
Trial 1 |
N=246 |
N=84 |
|
Trial 2 |
N=246 |
N=78 |
Jeuveau Prescribing Information
Were there any differences in how well the drug worked in clinical trials among sex, race and age?
- Sex: The majority of patients were women. The number of men was limited; therefore, differences in how well JEUVEAU worked in male patients compared to female patients could not be determined.
- Race: The majority of patients were White. The number of patients in other races were limited; therefore, differences in how well JEUVEAU worked among races could not be determined
- Age: The majority of patients were less than 65 years of age; therefore, differences in how well JEUVEAU worked among younger and older age groups could not be determined.
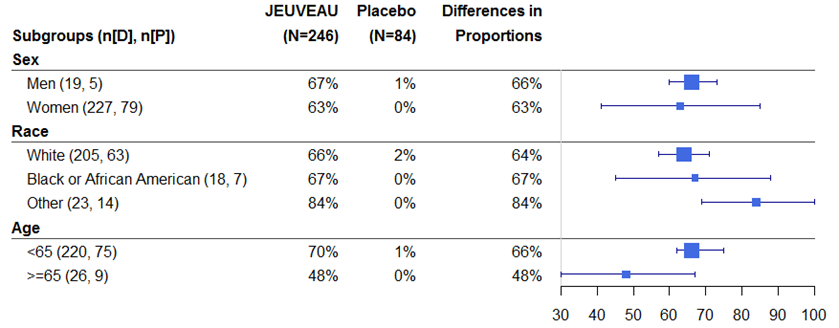
Were there any differences in how well the drug worked in clinical trials among sex, race, and age groups?
The tables below summarize efficacy results by sex, race, and age based on the proportion of patients achieving ≥2-grade improvement from baseline at maximum frown on Day 30.
Table 3. Trial 1: Subgroup Analysis of the Proportion of Patients Achieving ≥2-grade improvement in Glabellar Line Severity from Baseline at Maximum Frown on Day 30
N[D]= Subgroup sample size for JEUVEAU arm
N[P]= subgroup sample size for placebo arm
FDA Review
Table 4. Trial 2: Subgroup Analysis of the Proportion of Patients Achieving ≥2-grade improvement in Glabellar Line Severity from Baseline at Maximum Frown on Day 30
N[D]= Subgroup sample size for JEUVEAU arm
N[P]= subgroup sample size for placebo arm
FDA Review
What are the possible side effects?
Botulinum toxin products may spread from the area of injection to other areas and cause serious life-threatening side effects including swallowing and difficulty breathing that could lead to death. Spread of botulinum toxin products to other areas can also cause loss of strength and muscle weakness, double vision, blurred vision and drooping eyelids, hoarseness or change or loss of voice, trouble saying words clearly, and loss of bladder control.
Other serious side effects of botulinum toxin products include severe allergic reactions, and heart attack.
The most common side effects of JUVEAU are headache, upper respiratory tract infection, and drooping of eyelid.
What are the possible side effects (results of trials used to assess safety)?
The table below summarizes adverse reactions in patients with moderate to severe glabellar lines in the clinical trials.
Table 5. Adverse Reactions Reported at Higher Frequency (≥1%) in the JEUVEAU Group Compared to the Placebo Group
|
|
JEUVEAU
|
PLACEBO
|
|
Headache |
57 (12%) |
21 (13%) |
|
Upper Respiratory Tract Infection |
13 (3%) |
1 (1%) |
|
Eyelid Ptosis |
8 (2%) |
0 (0%) |
|
White Blood Cell Count Increase |
6 (1%) |
0 (0%) |
JEUVEAU Prescribing Information
Were there any differences in side effects among sex, race and age?
- Sex: The majority of patients were women. The number of men was limited; therefore, differences in the occurrence of side effects among male patients compared to female patients could not be determined.
- Race: The majority of patients were White. The number of patients in other races were limited; therefore, differences in the occurrence of side effects among races could not be determined.
- Age: The majority of patients were less than 65 years of age; therefore, differences in the occurrence of side effects among younger and older age groups could not be determined.
Were there any differences in side effects of the clinical trials among sex, race, and age groups?
The tables below summarize the occurrence of the most common adverse reaction, headache, by subgroup.
Table 6. Subgroup Analysis of Headache
|
Demographic Characteristic |
JEUVEAU |
Placebo |
|
Sex |
||
|
Men |
2/45 (4) |
1/13 (8) |
|
Women |
55/447 (12) |
20/149 (13) |
|
Race |
||
|
White |
45/420 (11) |
6/132 (12) |
|
Black or African American |
6/37 (16) |
1/13 (8) |
|
Asian |
0/7 (0) |
1/6 (17) |
|
Other |
6/28 (21) |
3/11 (27) |
|
Age Group |
||
|
Less than 65 years |
53/439 (12) |
19/147 (13) |
|
65 years and older |
4/53 (8) |
2/15 (13) |
Clinical Trial Data
WHO WAS IN THE CLINICAL TRIALS?
Who participated in the Clinical Trials?
The FDA approved JEUVEAU based on evidence from two clinical trials (Trial 1/NCT02334423 and Trial 2/NCT02334436) of 654 adult patients with moderate to severe glabellar lines (wrinkles between the eyebrows). The trials were conducted at 20 sites in the United States.
Figure 1 summarizes how many men and women were in the clinical trials.
Figure 1. Baseline Demographics by Sex
FDA Review
Figure 2 summarizes the percentage of patients by race in the clinical trials.
Figure 2. Baseline Demographics by Race
*Other includes American Indian or Alaska Native and Native Hawaiian or Other Pacific Islander
FDA Review
Table 1. Demographics of Trials by Race
|
Race |
Number of Patients |
Percentage of Patients |
|
White |
552 |
84% |
|
Black or African American |
50 |
8% |
|
Asian |
13 |
2% |
|
American Indian or Alaska Native |
2 |
Less than 1% |
|
Native Hawaiian or Other Pacific Islander |
1 |
Less than 1% |
|
Other |
36 |
5% |
FDA Review
Figure 3 summarizes the percentage of patients by age group in the clinical trials.
Figure 3. Baseline Demographics by Age
FDA Review
Who participated in the trials?
The table below summarizes demographics of the patients in the combined clinical trials.
Table 7. Baseline Demographics Trial 1 and Trial 2
|
|
Trial 1 |
Trial 2 |
Combined Clinical Trials |
||
|
JEUVEAU |
Placebo |
JEUVEAU |
Placebo |
(N=654) |
|
|
Sex |
|||||
|
Men |
19 (8) |
5 (6) |
26 (11) |
8 (10) |
58 (9) |
|
Women |
227 (92) |
79 (94) |
220 (89) |
70 (90) |
596 (91) |
|
Race |
|||||
|
White |
205 (83) |
63 (75) |
215 (87) |
69 (88) |
552 (84) |
|
Black or African American |
18 (7) |
7 (8) |
19 (8) |
6 (8) |
50 (8) |
|
Asian |
2 (1) |
4 (5) |
5 (2) |
2 (3) |
13 (2) |
|
American Indian or Alaska Native |
1 (<1) |
1 (<1) |
0 (0) |
0 (0) |
2 (<1) |
|
Native Hawaiian or Other Pacific Islander |
1 (<1) |
0 (0) |
0 (0) |
0 (0) |
1 (<1) |
|
Other |
19 (8) |
9 (11) |
7 (3) |
1 (1) |
36 (5) |
|
Age (years) |
|||||
|
Mean (SD) |
50.2 (11.8) |
50.4 (11.9) |
51.5 (11.5) |
50.4 (10.1) |
50.6 (11.3) |
|
Median |
52 |
51 |
53 |
49 |
51 |
|
Range |
22-81 |
23-74 |
21-81 |
18-71 |
18-81 |
|
Age Group |
|||||
|
< 65 years |
220 (89) |
75 (89) |
219 (89) |
72 (92) |
586 (90) |
|
> 65 years |
26 (11 ) |
9 (11) |
27 (11) |
6 (8) |
68 (10) |
|
Country |
|||||
|
United States |
246 (100) |
84 (100) |
246 (100) |
78 (100) |
654 (100) |
FDA Review
How were the trials designed?
The benefit and side effects of JEUVEAU were primarily evaluated in two clinical trials.
Both trials enrolled patients 18 to 81 years old with moderate to severe glabellar lines (wrinkles between the eyebrows). Patients received a single intramuscular injection of JEUVEAU or placebo at five sites within the muscles between the eyebrows.
The benefit of JEUVEAU was assessed by determining the proportion of patients achieving at least a 2-grade improvement of wrinkles between the eyebrows at maximum frown from baseline to Day 30. Improvement of wrinkles between the eyebrows at maximum frown was assessed independently by both the investigator and the patient using the Glabellar Line Scale (GLS). The GLS is a 4-point grading scale that assesses improvement (0=none, 1=mild, 2= moderate, 3=severe).
How were the trials designed?
The efficacy and safety of JEUVEAU were primarily evaluated in two randomized, double-blind, placebo-controlled trials.
Both trials enrolled patients ranging in age from 18 to 81 years old with moderate to severe glabellar facial lines at maximum frown. Patients were randomized to receive a single treatment with JEUVEAU or placebo intramuscularly. Patients received intramuscular injections (0.1 mL/injection site) of JEUVEAU or placebo at five sites, one in the procerus muscle and two in each corrugator supercilii muscle.
The primary efficacy endpoint was the proportion of patients achieving ≥2-grade improvement from baseline at maximum frown on Day 30. Improvement of glabellar line severity at maximum frown was assessed independently by both the investigator and the patient using the Glabellar Line Scale (GLS). The GLS is a 4-point grading scale (0=none, 1=mild, 2= moderate, 3=severe).
GLOSSARY
CLINICAL TRIAL: Voluntary research studies conducted in people and designed to answer specific questions about the safety or effectiveness of drugs, vaccines, other therapies, or new ways of using existing treatments.
COMPARATOR: A previously available treatment or placebo used in clinical trials that is compared to the actual drug being tested.
EFFICACY: How well the drug achieves the desired response when it is taken as described in a controlled clinical setting, such as during a clinical trial.
PLACEBO: An inactive substance or “sugar pill” that looks the same as, and is given the same way as, an active drug or treatment being tested. The effects of the active drug or treatment are compared to the effects of the placebo.
SUBGROUP: A subset of the population studied in a clinical trial. Demographic subsets include sex, race, and age groups.