Resources for Parents and Caregivers on Imported Infant Formula
The FDA has created resources for parents and caregivers to help explain some of the processes associated with, and results of, the agency’s efforts to help facilitate importing infant formula.
On May 16, 2022, FDA announced increased flexibilities for the importation of infant formula products, which have resulted in more than 520 million bottles worth of infant formula coming to the U.S.
Many of the imported products are, or will be soon, available through regular places to shop for infant formula, like major retailers, grocery stores and their online counterparts, as well as through company-specific websites.
Infant Formula Names to Know
Learn to recognize the labels of imported formula products you may see available at U.S. retailers now or coming soon. Click here for a few examples of imported infant formula you may be seeing.
Tips on Where to Find Products and Comparable Formulas
Although the supply of infant formula is steadily increasing, you may follow these tips to help find safe substitutes in the interim, including trying a new brand of formula (see list of comparable formulas) and talking to a pediatrician or health care provider about submitting an urgent request for specialized formula.
Video Gallery
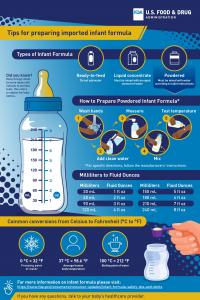
Learn the answers to frequently asked questions about imported infant formula, including questions about safety measures, how to prepare powdered formula and more.
Safety Review Process
Infant formula that is being imported to the U.S. undergoes a thorough review by the FDA. Review of the information provided by the companies includes looking at the:
- Ingredients
- Nutrient testing
- Manufacturing safety
- Allergen labeling
- Directions for preparation
Watch: Learn about what the FDA is doing to ensure the safety of imported infant formula.
FDA has been working collectively with federal partners, manufacturers and retailers to ensure confidence in infant formula that is safe and nutritious, including product coming in from other countries, is on shelves nationwide.
Additional Resources
- Infant Formula Information and Ongoing FDA Efforts to Increase Supply
- FDA Consumer Update: Infant Formula Supply
- Help Prevent Cronobacter Illness: Prepare and Store Powdered Infant Formula Safely
- Enforcement Discretion to Manufacturers to Increase Infant Formula Supplies
- HHS.gov/formula
- Powdered Infant Formula Recall: What to Know
- CDC Information on Cronobacter Infection and Infants