Outbreak Investigation of Salmonella: Onions (October 2023)
Do not eat, sell, or serve recalled diced onion products. FDA’s investigation is ongoing.
Product
Recalled Gills Onions-brand diced yellow onions, diced onions & celery, diced mirepoix, and diced red onions with use-by dates in August 2023.
FDA’s investigation is ongoing. Updates to this advisory will be provided as they become available.
Stores Affected
- Recalled Gills Onions were sold to foodservice and institutions nationwide and in Canada. The firm has directly notified foodservice customers who received recalled product.
- Recalled product was sold to retailers in Arizona, California, Idaho, Montana, Oregon, and Washington.
Symptoms of Salmonella Infection
Illness usually occurs within 12 to 72 hours after eating contaminated food and usually lasts four to seven days. Symptoms include diarrhea, fever, and abdominal cramps. Children younger than five, the elderly, and people with weakened immune systems are more likely to have severe infections.
Status
Ongoing
Recommendation
- Consumers should not eat, sell, or serve recalled onions and should check their freezers if they froze recalled products. Full descriptions of recalled products sold to retailers in AZ, CA, ID, MT, OR, and WA can be found in the recall announcement.
- Direct customers, including restaurants, institutions, and retailers will be contacted by the company and should check their refrigerators and freezers and throw away recalled onions.
- Contact your healthcare provider if you think you may have symptoms of a Salmonella infection after eating recalled onions.
Current Update
October 24, 2023
The U.S. Food and Drug Administration (FDA) and Centers for Disease Control and Prevention (CDC), in collaboration with state and local partners, are investigating an outbreak of Salmonella Thompson infections linked to fresh diced onions from Gills Onions of Oxnard, California.
Based on epidemiological data collected by CDC, 14 of 19 cases with information available reported exposure to fresh diced onions before becoming ill. Additionally, based on traceback data collected by FDA, FDA determined that onions processed at Gills Onions were available at points of service where people ate prior to becoming ill.
In response to this investigation, Gills Onions has voluntarily recalled diced yellow onion, diced onions & celery, diced mirepoix, and diced red onions.
The investigation is ongoing to determine the source of contamination and whether additional products are linked to illnesses. FDA will update this advisory should additional consumer safety information become available.
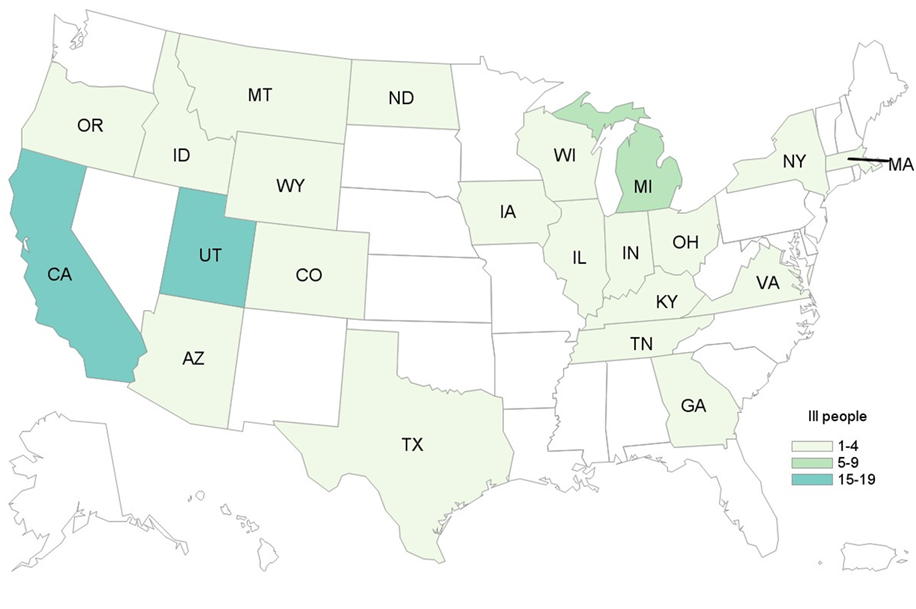
Case Count Map Provided by CDC
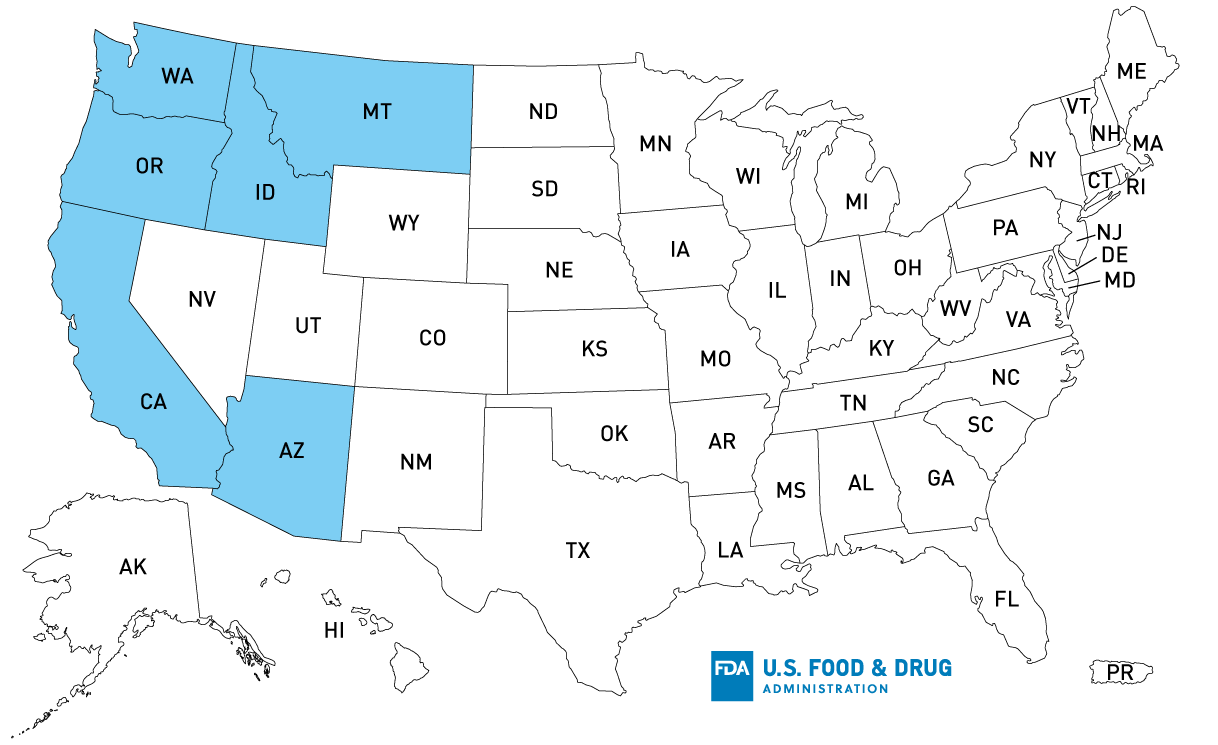
Map of U.S. Distribution of Recalled Onions Sold at Retail
Case Counts
Total Illnesses: 73
Hospitalizations: 15
Deaths: 0
Last Illness Onset: September 25, 2023
States with Cases: AZ, CA, CO, GA, ID, IL, IN, IA, KY, MA, MI, MT, NY, ND, OH, OR, TN, TX, UT, VA, WI, WY
Foodservice Product Distribution*: Nationwide and Canada
Retail Product Distribution*: AZ, CA, ID, MT, OR, WA
*Distribution has been confirmed for locations listed, but product could have been distributed further.
Product Images
Recalled Product
In response to this investigation, Gills Onions has voluntarily recalled diced yellow onion, diced onions & celery, diced mirepoix, and diced red onions. A full list of recalled retail products, including lot numbers, lot codes, and names of retail outlets can be found in the recall announcement.
International Distribution for Recalled Products
The recall impacts markets outside of the United States. Customer information provided by the firm shows that product was also distributed to foodservice customers to Canada. The firm has directly notified foodservice customers who received recalled product.
Who to Contact
Consumers who have symptoms should contact their health care provider to report their symptoms and receive care.
To report a complaint or adverse event (illness or serious allergic reaction), you can
- Call an FDA Consumer Complaint Coordinator if you wish to speak directly to a person about your problem.
- Complete an electronic Voluntary MedWatch form online.
- Complete a paper Voluntary MedWatch form that can be mailed to FDA.