FDA Drug Safety Communication: FDA recommends against use of Revatio (sildenafil) in children with pulmonary hypertension
Safety Announcement
Additional Information for Patients
Additional Information for Healthcare Professionals
Data Summary
[8-30-2012] The U.S. Food and Drug Administration (FDA) is recommending that Revatio (sildenafil) not be prescribed to children (ages 1 through 17) for pulmonary arterial hypertension (PAH; high pressure in the blood vessels leading to the lungs). This recommendation against use is based on a recent long-term clinical pediatric trial showing that: (1) children taking a high dose of Revatio had a higher risk of death than children taking a low dose and (2) the low doses of Revatio are not effective in improving exercise ability.1 Most deaths were caused by pulmonary hypertension and heart failure, which are the most common causes of death in children with PAH.
Facts about sildenafil
- A phosphodiesterase-5 inhibitor used to treat pulmonary arterial hypertension by relaxing the blood vessels in the lungs to reduce blood pressure.
- Also marketed in the prescription product Viagra, a drug for adult males with erectile dysfunction (inability to get or maintain an erection). At this time, FDA does not believe that this safety concern applies to the use of Viagra, given the differences in patient population and the different recommended dosing regimens.
Revatio has never been approved for the treatment of PAH in children, and in light of the new clinical trial information, off-label (not approved by FDA) use of the drug in pediatric patients is not recommended. The following new information is being added to the Revatio drug label:
- A new warning stating the use of Revatio is not recommended in pediatric patients
- Results of the Revatio trial in pediatric patients
Revatio is approved to improve exercise ability and delay clinical worsening of PAH in adult patients (WHO Group I). The current Revatio label recommends avoiding doses higher than 20 mg, given three times a day. The effect of Revatio on the risk of death with long-term use in adults is unknown; FDA is requiring the manufacturer of Revatio (Pfizer) to evaluate Revatio’s effect on the risk of death in adults with PAH.
Additional Information for Patients
- Do not change your Revatio dose or stop taking Revatio without talking to your health care professional.
- Discuss any questions you have about Revatio with your health care professional.
- Report any side effects you experience to the FDA MedWatch program using the information in the “Contact FDA” box at the bottom of the page.
Additional Information for Healthcare Professionals
- Use of Revatio, particularly chronic use, is not recommended in children. An unexpectedly higher risk of mortality was found in pediatric patients taking a high dose of Revatio when compared to pediatric patients taking a low dose. [see Data Summary]
- The maximum recommended dose of Revatio for adult patients with PAH is 20 mg three times a day.
- Report adverse events involving Revatio to the FDA MedWatch program, using the information in the “Contact FDA” box at the bottom of the page.
In a randomized, double-blind, multi-center, placebo-controlled, parallel-group, dose-ranging clinical trial, 234 patients with PAH, 1 to 17 years of age, were randomized to low-, medium-, or high-dose Revatio (sildenafil; administered three times per day) or placebo for 16 weeks of treatment. Most patients had mild to moderate symptoms at baseline. Actual doses administered were dependent on body weight.
The primary objective of the trial was to assess the effect of Revatio on exercise capacity as measured by the Cardiopulmonary Exercise Test (CPET) in patients who were developmentally able to perform the test (n = 115). Administration of Revatio did not result in a statistically significant improvement in exercise capacity in those patients.
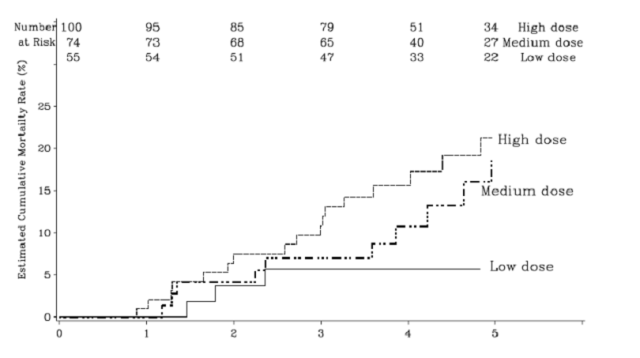
After completing the 16-week controlled clinical trial, patients randomized to Revatio remained on the same dose of Revatio; patients originally randomized to placebo were re-randomized to low-, medium-, or high-dose Revatio. After all patients completed 16 weeks of follow-up in the controlled clinical trial, the blind was broken and doses were adjusted as clinically indicated. Patients were followed for a mean of 3 years (range 0 to 7 years). Mortality during the long-term clinical trial, as a function of originally assigned dose, is shown in the figure below.
Figure: plot of mortality in the pediatric clinical trial as a function of Revatio dose.
A direct dose-related effect on mortality was observed with the highest dose having the worst outcome. The hazard ratio for high dose compared to low dose was 3.5 (p=0.015). Deaths were first observed after about 1 year, then occurred at fairly constant rates within each group. Causes of death were typical of patients with pulmonary hypertension.
In light of these risks, the use of Revatio is not recommended in children. A new warning against the use of Revatio in pediatric patients is being added to the Revatio drug label.
References
- Barst RJ, Ivy DD, Gaitan G, et al. A randomized, double-blind, placebo-controlled, dose-ranging study of oral sildenafil citrate in treatment-naïve children with pulmonary arterial hypertension. Circulation 2012;125:324-334.
Related Information
- FDA Drug Safety Podcast: FDA recommends against use of Revatio in children with pulmonary hypertension
- Sildenafil (marketed as Viagra and Revatio) Information
- Comunicado de la FDA sobre la seguridad de los medicamentos La FDA recomienda que no se use Revatio en niños con hipertensión pulmonar
- FDA clarifies warning about Pediatric Use of Revatio (sildenafil) for Pulmonary Arterial Hypertension