Boston Scientific Recalls EMBLEM S-ICD Subcutaneous Electrode (Model 3501) Due to Risk of Fractures
The FDA has identified this as a Class I recall, the most serious type of recall. Use of these devices may cause serious injuries or death.
Recalled Products
EMBLEM S-ICD Subcutaneous Electrode
- Model Number: 3501
- Manufacturing Dates: March 2016 to present
- Distribution Dates: June 2017 to present
- Devices Recalled in the U.S.: 19,919
- Date Initiated by Firm: December 2, 2020
Device Use
The EMBLEM S-ICD Subcutaneous Electrode Model 3501 is part of the Boston Scientific S-ICD System. The Boston Scientific S-ICD is an implantable cardioverter defibrillator that is intended to provide electrical shock to stop dangerously fast heart rhythms and pacing for a short time after shocks if needed. These cardiac devices are implanted under the skin in the upper chest area.
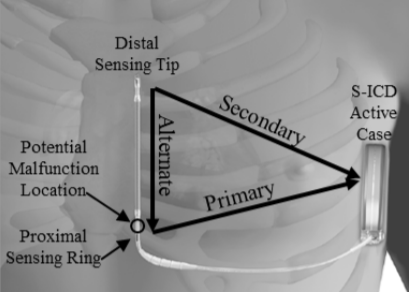
Fig. 1: Picture of the EMBLEM S-ICD Subcutaneous Electrode with an arrow showing where separation usually happens (distal to the proximal sense ring).
Reason for Recall
Boston Scientific is recalling the EMBLEM S-ICD Subcutaneous Electrode because of increased risk of fractures at a specific point (distal to the proximal sense ring) shown in Figure 1. If the device fractures during use, it could become unable to deliver therapy to slow very fast heartbeats from cardiac arrest (tachycardia). A failed device may cause serious adverse events. Examples include injury or death if cardiac arrest cannot be treated or need for additional surgery to replace failed devices.
There have been 27 complaints about this device issue and 26 reports of serious injuries. One death has been reported.
Who May be Affected
- Health care providers using the affected Boston Scientific device
- Patients who have procedures with the affected Boston Scientific device
What to Do
On December 2, 2020, Boston Scientific sent an Important Medical Device Advisory letter to all affected customers with recommendations for prompt identification of devices at risk for electrode body fracture. Recommendations were offered to help physicians and patients evaluate the risks of using affected devices compared to replacing them. Care for recalled devices at risk for failure were provided:
- Enroll and monitor patients through LATITUDE remote monitoring to detect any alerts or artifacts on the devices in between office device checks.
- Ask patients to do weekly remote checks
- Perform a system follow-up every three months by remote or in-office checks.
- Assess sensing performance in-clinic if any of the following is observed:
- Non-physiologic, mechanical artifacts
- If artifacts are found, this may be a sign of an electrode body fracture.
- High electrode impedance alerts.
- Non-physiologic, mechanical artifacts
- Capture all sensing vectors that may indicate onset of electrode body fracture such as:
- Cardiac signals on the S-ECGs of the Primary and Secondary sensing vector that look nearly identical
- Flatline S-ECGs in the Alternate sensing vector
- Review stored devices for artifacts, as this may be a sign of potential electrode body fracture
- Assess sensing performance in-clinic if any of the following is observed:
- Perform a chest radiography of the entire electrode length if there is a suspicion of a break.
- In the absence of any indications of electrode fracture, surveillance X-rays are not recommended.
- Demonstrate the device beeper to the patient during the next office visit.
- For patients not monitored by LATITUDE, repeat the beeper demonstration following any MRI scan, as strong magnetic fields may cause permanent loss of beeper volume
- Remind all patients to quickly contact their physician if beeping tones are heard from their device or if a shock is delivered.
- Evaluate the risk for life-threatening harm due to an electrode body fracture. This is greatest for patients who:
- Have a history of life-threatening ventricular arrhythmias such as secondary prevention
- Are unable to be reliably followed remotely or in person every three months
- Are not monitored via LATITUDE and are unable to hear beeping tones
- Replace any electrode that is shown to have breaks with signs like artifacts, high impedance alert, or X-ray results.
- Routine replacement of an electrode without evidence of breaking is not recommended
- Return explanted devices to Boston Scientific
- Update medical Records with this letter for each patient with an EMBLEM S-ICD Subcutaneous Electrode (Model 3501).
Contact Information
Customers who have questions about the notification should contact their local sales representative or the Boston Scientific Technical Services team by phone at (800)-CARDIAC (227-3422) or by emailing [email protected].
Additional Resources
How do I report a problem?
Health care professionals and consumers may report adverse reactions or quality problems they experienced using these devices to MedWatch: The FDA Safety Information and Adverse Event Reporting Program using an online form, regular mail, or FAX.