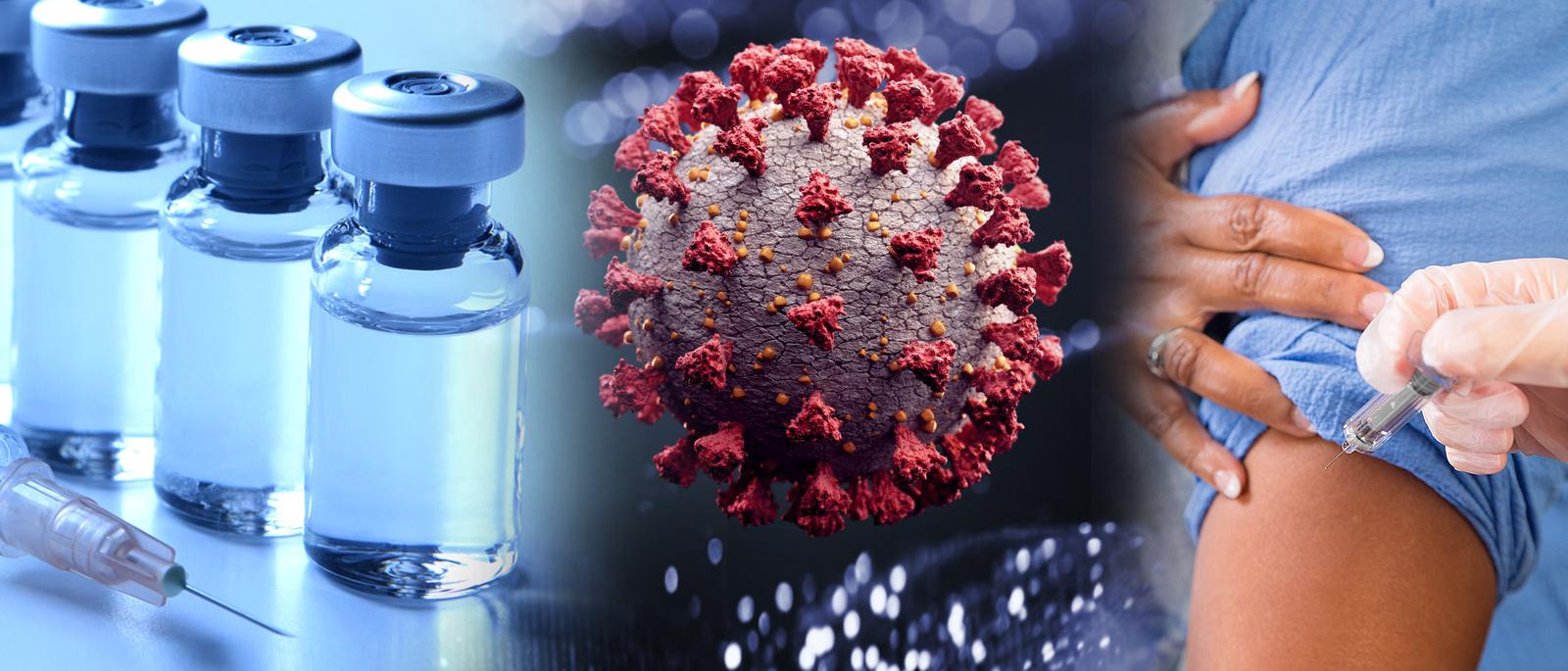
Virtual Press Conference: FDA to Discuss Pfizer-BioNTech COVID-19 Vaccine EUA for Children 5 Through 11
Acting FDA Commissioner Dr. Janet Woodcock and FDA's Center for Biologics Evaluation and Research Director Dr. Peter Marks discuss the FDA expanding the emergency use authorization of Pfizer-BioNTech COVID-19 vaccine to include children 5 through 11
Watch press conferenceFeatured Topics

New Era of Smarter Food Safety
A new approach to food safety that recognizes and builds on the progress made in the past but looks towards what processes and tools will be needed for the future.

Fostering Drug Competition
Increasing the availability of generic drugs helps to create competition in the marketplace, helping make treatment more affordable and increasing access to healthcare for more patients.

Combating the Opioid Crisis
FDA takes actions to combat prescription opioid abuse.
Recalls, Market Withdrawals, & Safety Alerts
The FDA provides information gathered from press releases and other public notices about certain recalls of FDA-regulated products. Not all recalls have press releases or are posted on this website
Oct 29
Dole Fresh Vegetables Announces Precautionary Limited Recall of Garden Classic Salads for Possible Contamination with Listeria monocytogenes
Food & Beverages
Oct 27
Purina® Animal Nutrition Voluntarily Recalling Limited Lots of Purina Cattle and Wildlife Feed Due to Elevated Urea Levels
Animal & Veterinary
Oct 27
Bobo’s Issues a Voluntary Allergy Alert on Undeclared Peanuts in Almond Butter Bars
Food & Beverages
FDA VOICES: PERSPECTIVES FROM FDA EXPERTS
How CDRH’s Digital Transformation Initiative Will Strengthen the Premarket Review Program
To meet our public health mission, it is critical that FDA continues to evolve to help speed innovations that make medical products safer and more effective.

FDA’s Advanced Manufacturing Initiatives Helping to Provide Quality Human Drugs for Patients
FDA has established numerous initiatives, including the development of a research program to better understand the science of advanced manufacturing.

Advancing the Development of Innovative Veterinary Products
FDA takes a proactive approach to provide timely and efficient review of innovative medical products for animals.
Recently Published Guidances
Oct 29
Reporting Amount of Listed Drugs and Biological Products Under Section 510(j)(3) of the Federal Food, Drug, and Cosmetic Act; Draft Guidance for Industry
Drugs
Oct 29
Reporting Amount of Listed Drugs and Biological Products Technical Conformance Guide
Drugs
Oct 21
Data Standards for Drug and Biological Product Submissions Containing Real-World Data
Biologics, Drugs