VIP: Veterinary Innovation Program
for Certain New Animal Drug Applications for Intentional Genomic Alterations in Animals and Animal Cell, Tissues, and Cell- or Tissue-Based Products
On this page:
What is the VIP?
Who Can Participate in the VIP?
How will I know if my product can be in the VIP?
What are the Benefits of the VIP?
Can my product’s qualification for the VIP be withdrawn?
What is the VIP?
The FDA Center for Veterinary Medicine’s (CVM) Veterinary Innovation Program (VIP) is for certain intentional genomic alterations (IGA) in animals and animal cells, tissues, and cell- or tissue-based products (ACTPs) seeking FDA approval of a new animal drug application (NADA). The goal of the VIP is to facilitate advancements in development of innovative animal products by providing greater certainty in the regulatory process, encouraging development and research, and supporting an efficient and predictable pathway to approval for ACTPs and IGAs in animals.
The VIP offers benefits including extensive interactions between CVM and the sponsor (you). These interactions can increase the predictability of the regulatory pathway, facilitate a lower number of review cycles, and reduce the overall time to approval. Continuing interactions and collaboration with CVM throughout the approval process maximizes the ability to address potential challenges in product development, leverage data from multiple sources, minimize the number of studies conducted to support approval, enhance communication in the review process, and maintain an understanding of current scientific and regulatory advancements.
Who Can Participate in the VIP?
Sponsors of IGAs in animals or ACTPs that provide a benefit to human health, animal health, animal well-being (e.g. husbandry improvements), or enhanced food production may participate in the VIP. For purposes of this program, an IGA in an animal is an intentional alteration to an animal's DNA that has been made using modern molecular technologies and an ACTP is one that contains, consists of, or is derived from cells or tissues that are intended for implantation, transplantation, infusion, or transfer into an animal recipient. If you are unsure if your product is an IGA or ACTP over which FDA has jurisdiction, contact us at askcvm@fda.hhs.gov.
Submission quality is critical to achieving efficiency in the approval process. The VIP includes benefits intended to ensure you have the necessary tools to generate quality submissions.
How will I know if my product can be in the VIP?
We recommend you contact CVM as early as possible in the development process if you are interested in participating in the VIP for your product.
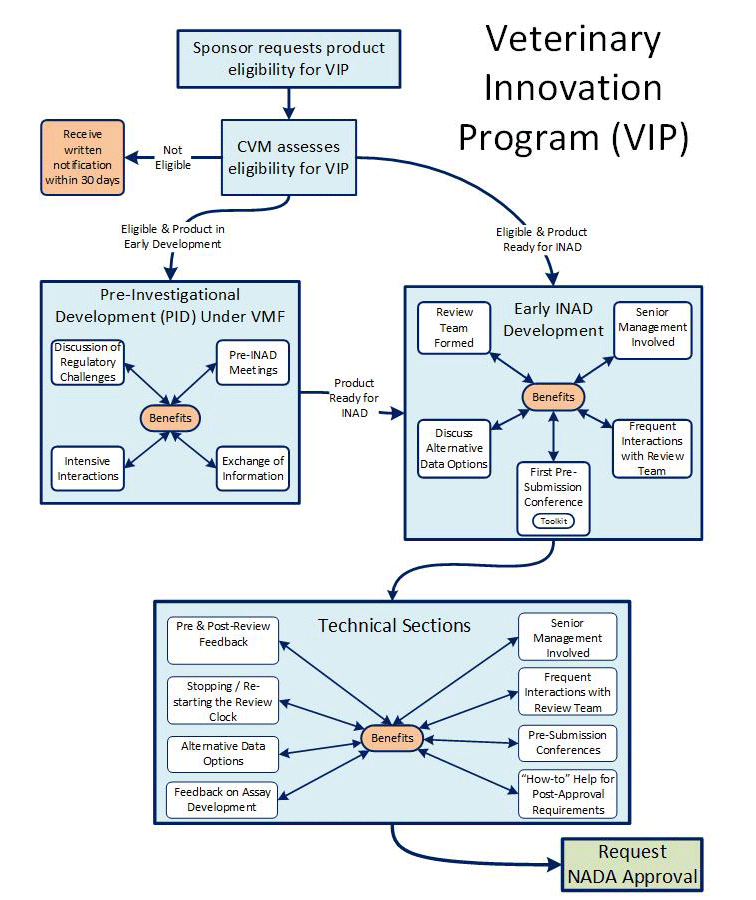
Your ACTP or IGA qualifies for the VIP if you have not heard from us in writing that it does not qualify within 30 days of your initial request to establish an investigational new animal drug file (INAD) or veterinary master file (VMF) (contact CVM about which type of file is appropriate for your product). If you receive written notice that your product does not qualify for the VIP, and you believe that it qualifies, you should contact CVM and discuss what additional information you should provide that will explain how your product is either an ACTP or an IGA in an animal that provides a benefit to human health, animal health, animal well-being (e.g. husbandry improvements), or food production. CVM will review the information in your INAD or VMF relating to the product description, proposed claim or indication, and mechanism of action in determining whether the product qualifies for the VIP.
What are the Benefits of the VIP?
The VIP provides intensive assistance and enhanced review efficiency for developers of ACTPs and IGAs in animals.
- Intensive Interaction
Under the VIP, we believe it is important to begin working with you early in the development process. We will meet with you (through pre-INAD meetings, multiple pre-submission conferences, as well as meetings prior to and after individual submissions) to identify as early as possible the critical risk questions and to provide advice on a strategy for generating data that can support an eventual approval. During these meetings, we will provide feedback on proposals you give us prior to the meeting that describe the information you plan to include in each technical section. We will also provide assistance with determining appropriate methods to address unique challenges such as product identification, characterization, durability, and demonstrating comparability (e.g. between cell lines or alterations).
We encourage you to use both formal and informal communication with CVM to address individual development questions as they arise.
CVM Project Managers will have frequent communications with you throughout the approval process. These interactions may begin during the Pre-Investigational Development (PID) stage and continue throughout the approval process. Project Managers will assist you in understanding the regulatory process, planning for submissions, and ensuring you have the information necessary to prepare high quality submissions. Project Managers are your overall point of contact for the project. The review team lead (as described below) will serve as your point of contact for all scientific or submission-specific technical questions.
- Review Team
For each product, CVM will form a review team consisting of a representative from each relevant area of expertise (e.g. human food safety; environmental safety; target animal safety; effectiveness; biostatistics; chemistry, manufacturing and controls; animal bioengineering and cellular therapies (ABCT); and surveillance and compliance).
Each review team will include a lead from the ABCT Team who will facilitate the review process and serve as a scientific liaison between the cross-disciplinary members of the review team. The review team lead is your primary scientific point of contact, arranges formal and informal meetings with you, coordinates responses to your questions, coordinates and communicates review-related information to you, and ensures timely and consistent communication with you. The review team lead will work in close communication with the project manager and will lead collaboration between you and the review team to identify and address product development challenges.
In the event the review team determines that a submission is incomplete, the review team lead will communicate this information via email or telephone to you following completion of the submission review but in advance of providing the official decision to you in a letter. This advance information on the status of the submission is intended to assist you with planning and allow for efficient scheduling of a post-review feedback meeting (see Section d below). However, we will not specify the deficient items in the submission until we finalize the review decision and provide our comments in the letter.
- Pre-Investigational Development
Sponsors may contact us and engage in discussions in the PID phase. The PID phase refers to the early stages of research and development that typically take place prior to determining the precise product and indication that will be the subject of an NADA. PID, therefore, usually takes place prior to establishment of an INAD. PID allows for early interactions with FDA that may relate to scientific information regarding a novel product, challenges associated with product development, investigational use of a novel product for discovery and proof-of-concept studies, exchange of other information intended to facilitate research and development, or other interactions that will help define the pathway to approval. If your product has a proposed intended use or claim that may ultimately require a separate approval from another FDA Center, we will include the relevant center in these early discussions, as needed.
To engage in interactions with us in the PID phase, we recommend you establish a VMF rather than an INAD. Unlike an INAD, the establishment of a VMF is not subject to user fees.
- Pre-Review Feedback
You may request input on the organization and general contents of a technical section submission prior to formally providing the submission to CVM. Pre-review feedback is intended to assist sponsors with products in the VIP that are new to the FDA regulatory process and unfamiliar with FDA’s format and content expectations for technical section submissions and is intended to facilitate an efficient review process.
During pre-review, we will provide high level feedback sufficient to identify data gaps and address your specific questions regarding overall submission content. For example, we will identify missing data or items requiring justification. We will not address questions regarding acceptability of the submission contents or provide detailed lists of comments.
Requests for pre-review feedback should include a table of contents or outline for the submission as well as an indication of the level of detail you intend to provide for each section (e.g., final study report for study x, literature regarding item y, validation data generated from x). Requests for pre-review feedback should be submitted at least 30 days prior to submission of the technical section.
Requests for pre-review feedback should not include product development questions or questions relating to clarification of comments previously transmitted to the sponsor in incomplete or non-concurrence letters or meeting minutes. These questions should be directed to the review team lead in a separate communication.
Pre-review feedback is intended to prevent rejection of submissions due to incomplete information and to ensure you have an adequate understanding of CVM expectations to generate high-quality submissions. In the event that a sponsor repeatedly requests pre-review feedback for the same type of submission or question, or repeatedly submits poor quality submissions despite assistance through pre-review feedback, we may elect to cease providing pre-review feedback until the sponsor asks new questions or submission quality improves.
- Post-Review Feedback
CVM will offer post-review feedback to clarify comments we have sent you in response to an incomplete protocol, technical section, or other substantial submission. Post-review feedback is also intended to facilitate communication and understanding of scientific challenges facing product development.
Once we determine that a protocol, technical section or other substantial submission may be incomplete, the review team lead will schedule a teleconference with you to be held shortly after you receive the incomplete letter. The post-review feedback meeting should be limited to a discussion of our comments in an incomplete letter. We recommended that you inform CVM of the comments you would like to discuss prior to the meeting date to facilitate productive communication. We will not provide meeting minutes for post-review feedback meetings, as the meetings are meant to clarify our previously transmitted comments. If you wish to discuss new proposals or questions, you should schedule a separate meeting.
You may also request “lessons learned” meetings following product approval. At a “lessons learned” meeting CVM will provide feedback on what worked well with the approval process and will offer suggestions for improvement regarding submission quality. You will also have the opportunity to provide your feedback about the overall process and interactions with CVM that occurred during the approval process.
- Senior Manager Involvement
Products in the VIP will receive a high level of attention and organizational commitment involving CVM senior leadership. The review team will inform the Office of New Animal Drug Evaluation (ONADE) senior leadership of meetings and review decisions that impact product development and approval and, where needed, the senior leadership may advise the review team in addressing regulatory challenges impacting product development and approval. Senior leadership will also ensure that the review team has the appropriate resources and support to complete reviews of products in the VIP in a timely fashion and will keep other Center and FDA management apprised of the project status and progress towards approval.
- Tool Kit
We will provide you with materials that provide a head-start in preparing quality submissions with data and information to satisfy the requirements for each technical section. This toolkit may include materials such as an overview of the approval process, a list of relevant guidance documents and other references, and access to product-specific, question-based eSubmitter templates to further guide submissions.
- "How-To” Help for Meeting Post-Approval Obligations
We will work with you in the pre-approval stage to ensure a smooth transition to complying with post-approval requirements. As a VIP product nears approval, you may request a meeting with us to discuss post-approval requirements. We will provide hands on assistance with electronic registration and listing (21 CFR Part 207) and submission of post-approval reports (21 CFR 514.80). We will also help you determine the appropriate type of post-approval submission to file if you make any post-approval manufacturing changes to your product (e.g., what would be considered a moderate change that would require submission of a supplement 30 days before distributing the product made with the change (21 CFR 514.8(b)(3)) versus a major change that would require FDA approval of a supplement prior to distribution of the product made with the change (21 CFR 514.8(b)(2))).
- Identification and Assay Methods
Where required in an NADA, analytical methods can be scientifically challenging to develop for IGAs in animals and ACTP products. In these cases we may offer technical advice for the development and validation of identification and assay methods. For example, for identification methods utilized to detect genetic alteration(s) to an animal and to demonstrate that the alteration(s) to the animal has not changed from that which FDA determined to be safe and effective, or for in vitro potency assays supporting in vivo effectiveness, we may provide feedback on the proposed method and validation process.
- Stopping the Review Clock
For products in the VIP, we may stop the clock during the review of major technical section submissions in order to allow the sponsor time to receive and address feedback. Under the Animal Drug User Fee Act (ADUFA) review timelines, each technical section submission that will support an NADA approval has a 180-day review time. Ordinarily, when CVM determines a technical section submission is incomplete, we send the sponsor a letter informing them of this determination and the submission is closed out and removed from the queue. When the sponsor submits a revised version of the technical section, the 180-day clock starts anew. For major technical section submissions submitted under the VIP, CVM may stop the review clock to provide preliminary feedback to you regarding missing or unacceptable elements of the submission without closing out the submission or removing it from the queue. When you submit an amendment to address the deficiencies, the clock restarts. You may also choose to allow the clock to continue and receive a detailed incomplete letter once the review is completed. Stopping the review clock can shorten the time to address each technical section in support of NADA approval. Note, though, that if a submission is missing major pieces of information (e.g. genotypic characterization data, animal health data, etc.) that impact the scientific evaluation of that submission, we may choose to issue an incomplete letter rather than stopping the review clock.
- Alternative Data Options
To help facilitate development of VIPs, we may, where appropriate, accept alternative strategies for generating data or submission of different types of data in support of an approval. Examples of alternative types of data that we may accept include data from a limited number or generations/lineages of animals, data intended to support multiple cell lines/batches or genetic alterations in a single approval, data generated through collaboration across institutions, data from a limited number of animals supporting expanded indications, or data from alternative study designs.
We may also participate in brainstorming meetings to develop alternative strategies for data generation in support of an approval. For these meetings, you should identify areas for discussion, and we will actively participate in the development of these strategies. For example, we may provide, for discussion, alternative approaches to statistical analyses or risk-based plans for evaluation of safety. By participating in these meetings, we hope to reduce the potential for regulatory and scientific barriers that may impact approval process efficiency.
Our acceptance of an alternative approach to generating data depends on the type of ACTP or IGA and its indication/intended use. You should request meetings with us early in the development process to discuss these alternative strategies.
Can my product’s qualification for the VIP be withdrawn?
In the event that you repeatedly submit poor quality submissions for a product despite assistance received through the VIP, we may elect to withdraw your product’s qualification for this program. Similarly, your product may not be eligible for certain VIP benefits in some circumstances. For example, stopping the review clock is intended to address scientific issues related to review of novel products. It is not intended to rehabilitate poor quality submissions. A submission is only eligible to stop the review clock if the submission quality is acceptable.